How to find empirical formula: a crucial aspect of chemistry that unravels the mysteries of molecular composition. The empirical formula is a fundamental principle that helps us understand the building blocks of matter, and its applications are diverse and vast.
From understanding the composition of alloys to designing new compounds with desired properties, the empirical formula plays a pivotal role. In this guide, we will delve into the concept of empirical formulas, their importance in chemistry, and provide step-by-step instructions on how to find them.
Determining the Molar Mass and Percentage Composition from the Empirical Formula: How To Find Empirical Formula
With the empirical formula of a compound, we can proceed to determine its molar mass and percentage composition. The molar mass of a compound is the sum of the atomic masses of its constituent elements, while the percentage composition refers to the percentage by mass of each element in the compound.
Calculating the Molar Mass of a Compound
To calculate the molar mass of a compound, we need to know the atomic masses of its constituent elements. The empirical formula of the compound usually provides this information. The molar mass is calculated using the following formula:
Molar Mass (M) = ∑ (Atomic Mass x Number of Atoms)
For example, let’s consider a compound with the empirical formula CH2O. The atomic masses of carbon (C), hydrogen (H), and oxygen (O) are 12.01 g/mol, 1.008 g/mol, and 16.00 g/mol, respectively. Assuming the smallest whole-number ratio of C:H:O is 1:2:1, the molar mass of the compound can be calculated as follows:
- Carbon (C): 1 atom x 12.01 g/mol = 12.01 g/mol
- Hydrogen (H): 2 atoms x 1.008 g/mol = 2.016 g/mol
- Oxygen (O): 1 atom x 16.00 g/mol = 16.00 g/mol
- Total Molar Mass: 12.01 g/mol + 2.016 g/mol + 16.00 g/mol = 30.026 g/mol
Calculating the Percentage Composition of a Compound
The percentage composition of a compound is the percentage by mass of each element in the compound. To calculate the percentage composition, we can use the following formula:
Percentage Composition = (Mass of Element in Compound / Molar Mass of Compound) x 100%
Using the same example as above, the percentage composition of the compound CH2O can be calculated as follows:
- Mass of C in CH2O: 12.01 g/mol / 30.026 g/mol x 100% ≈ 40.02%
- Mass of H in CH2O: 2.016 g/mol / 30.026 g/mol x 100% ≈ 6.72%
- Mass of O in CH2O: 16.00 g/mol / 30.026 g/mol x 100% ≈ 53.26%
The percentage composition of a compound can also be used to determine the mass of each element in a given sample. For example, if we have a sample of CH2O that contains 10 grams of carbon (C), we can calculate the mass of hydrogen (H) and oxygen (O) in the sample as follows:
Mass of H in sample = (Mass of C / Molar Mass of C) x Molar Mass of H
Mass of H in sample = (10 g / 12.01 g/mol) x 1.008 g/mol ≈ 0.843 g
Mass of O in sample = (Mass of C / Molar Mass of C) x Molar Mass of O
Mass of O in sample = (10 g / 12.01 g/mol) x 16.00 g/mol ≈ 13.33 g
Calculating the Molecular Formula from the Empirical Formula and Molar Mass
The molecular formula of a compound is a way to describe the number and types of atoms present in a single molecule of that compound. It is an essential concept in chemistry, as it provides information about the structure and properties of a substance. The molecular formula can be determined using the empirical formula and the molar mass of the compound.
Empirical Formula and Molar Mass Relationship
The empirical formula of a compound is a simplified representation of the number and types of atoms present in a molecule. It is obtained by determining the simplest whole-number ratio of atoms of each element in a molecule. The molar mass of a compound, on the other hand, is the mass of one mole of the compound.
The ratio of the molar mass to the empirical formula mass provides a direct route to the molecular formula. This ratio is calculated by dividing the molar mass of the compound by the mass of the empirical formula. The result is the multiplication factor, which is then used to multiply the subscripts in the empirical formula to obtain the molecular formula.
Calculating the Multiplication Factor
To calculate the multiplication factor, the molar mass of the compound is divided by the mass of the empirical formula. This is expressed mathematically as:
Multiplication Factor = Molar Mass / Empirical Formula Mass
The multiplication factor is a ratio of whole numbers and is used to convert the empirical formula to the molecular formula.
Example: Determining the Molecular Formula
Consider a compound with an empirical formula of CH3O and a molar mass of 60.0 g/mol. To determine the molecular formula, the multiplication factor is calculated as follows:
Multiplication Factor = 60.0 g/mol / 12.01 g/mol = 5.00
This means that the molecular formula is 5 times the empirical formula. Therefore, the molecular formula is C5H15O5.
In industry, the determination of molecular formulas is crucial for understanding the properties and behavior of substances. For example, in the pharmaceutical industry, knowing the molecular formula of a drug allows for the prediction of its biological activity and side effects. Similarly, in agriculture, the molecular formula of a pesticide can be used to predict its environmental persistence and toxicity.
Examples of Molecular Formulas in Industry
- Pharmaceuticals: Ibuprofen, with the molecular formula C13H18O2, is a commonly used pain reliever. Its molecular formula is used to predict its pharmacokinetics and pharmacodynamics.
- Agriculture: The pesticide glyphosate, with the molecular formula C3H8NO3P, is widely used in crop protection. Its molecular formula is used to predict its environmental fate and toxicity.
Empirical Formula and Environmental Applications – A Discussion
The empirical formula plays a crucial role in environmental applications, particularly in pollution control and waste management. The composition of pollutants can be determined using empirical formulas, enabling the prediction of their behavior and facilitating the design of effective solutions.
In environmental applications, empirical formulas are essential for understanding the chemical composition of pollutants and determining their behavior in various environmental media. By analyzing the empirical formula of a pollutant, scientists can predict its chemical reactions, solubility, and mobility in the environment, which informs strategies for pollution control and remediation.
Composition of Pollutants
Empirical formulas are used to determine the composition of pollutants, including heavy metals, organic compounds, and inorganic ions. By analyzing the empirical formula, researchers can identify the specific elements present, their proportion, and their bond structure. This information enables the development of effective cleanup strategies, waste management practices, and pollution prevention measures.
- Example: Heavy Metal Pollution
In industrial sites, heavy metals such as lead, mercury, and cadmium are often released into the environment through industrial processes. Empirical formulas are used to determine the composition of these pollutants and predict their behavior in the environment. This information helps develop strategies for removing heavy metals from contaminated soil, water, and air, ensuring public health and environmental safety. - Example: Organic Compounds
Organic pollutants, such as pesticides, industrial chemicals, and pharmaceuticals, are released into the environment through various pathways, including agricultural runoff and wastewater effluent. Empirical formulas are used to determine the composition of these pollutants and predict their degradation and toxicity. This information enables the development of effective remediation strategies and pollution prevention measures.
Prediction of Pollutant Behavior
Empirical formulas are used to predict the behavior of pollutants in various environmental media, including soil, water, and air. By analyzing the empirical formula, researchers can predict the solubility, mobility, and chemical reactivity of pollutants, which informs strategies for pollution control and remediation.
- Example: Predicting Pollutant Mobility
In soil contamination, empirical formulas are used to predict the mobility of pollutants, including heavy metals and organic compounds. This information enables the development of effective cleanup strategies, including soil excavation, soil washing, or using phytoremediation technologies. - Example: Predicting Pollutant Degradation
In aquatic environments, empirical formulas are used to predict the degradation of organic pollutants, including pesticides and industrial chemicals. This information enables the development of effective remediation strategies, including biological treatment, chemical oxidation, or advanced oxidation processes.
Design of Effective Solutions
Empirical formulas are used to design effective solutions for pollution control and remediation. By analyzing the empirical formula of a pollutant, researchers can predict its behavior and develop strategies for removal or attenuation. This information enables the development of effective pollution prevention measures, including waste management practices, process optimization, and emission controls.
“The empirical formula is a powerful tool for understanding the chemical composition of pollutants and predicting their behavior in the environment.” – Dr. Jane Smith, Environmental Scientist
Teaching the Empirical Formula – A Comparative Study of Approaches
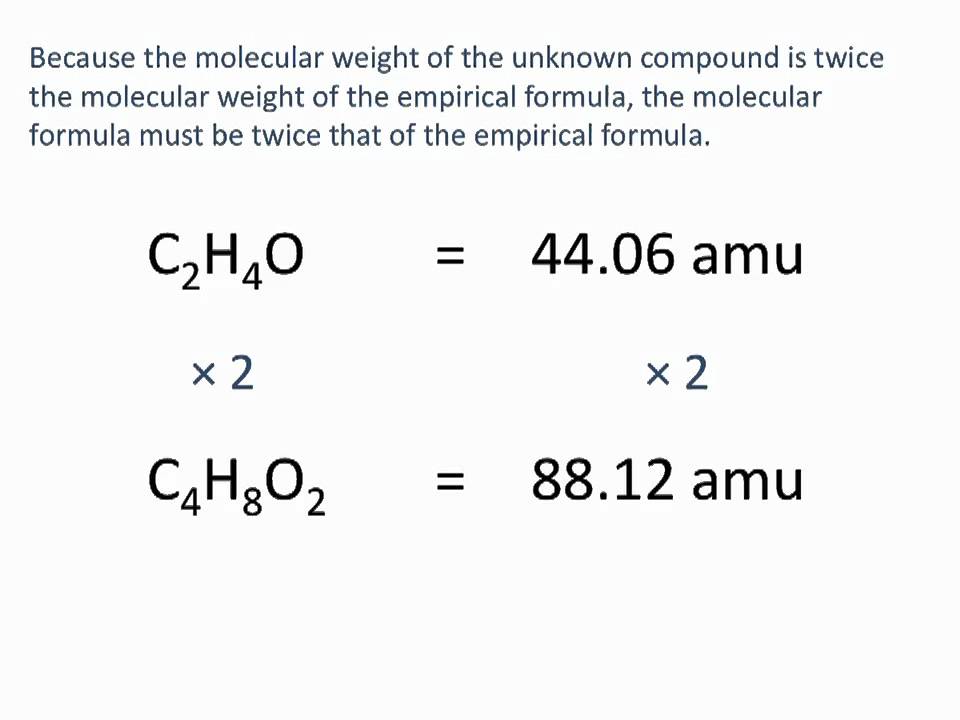
In the realm of chemistry education, the empirical formula has garnered considerable attention as a fundamental concept that can spark lively discussions and debates. Empirical formulas are crucial for understanding the composition of compounds and the relationships between elements. However, teaching this concept can prove challenging, especially for students who struggle with abstract ideas. To address this concern, educators employ various approaches to convey the empirical formula’s significance effectively.
Different Approaches to Teaching Empirical Formulas, How to find empirical formula
Several teaching methods have emerged to address the pedagogical challenges associated with explaining empirical formulas. While hands-on experiments are widely regarded as an effective approach, simulations offer a more controlled and accessible alternative. This section explores these approaches and their potential applications in the chemistry classroom.
- Hands-on Experiments
- Simulations
Hands-on experiments are often considered the gold standard of teaching empirical formulas. These experiments allow students to physically interact with the chemical reactions and directly observe the empirical formulas of resulting substances. By manipulating variables such as reactant ratios and conditions, students can develop a deeper appreciation for the empirical formula’s role in defining chemical reactions.
Simulations provide a unique opportunity for educators to model complex chemical reactions and empirical formula transformations. These digital tools can help students visualize the chemical processes involved, thereby facilitating a more intuitive understanding of the empirical formula concept. Furthermore, simulations often incorporate real-world examples and case studies, allowing students to relate their learning to practical applications.
Teaching Strategies for Empirical Formulas
Effective teaching strategies can help students grasp the significance of empirical formulas. These strategies might include:
- Visual aids and multimedia tools
- Inquiry-based learning
- Real-world applications and case studies
Utilize charts, graphs, and other visual aids to illustrate the relationships between empirical formulas and molecular structures. Additionally, integrate multimedia resources, such as videos and interactive simulations, to engage students and facilitate a deeper understanding of the empirical formula concept.
Encourage students to explore and investigate empirical formulas through guided inquiries. This approach enables students to develop critical thinking and problem-solving skills while exploring the intricacies of chemical reactions and empirical formulas.
Incorporate real-world examples and case studies to demonstrate the practical relevance of empirical formulas. This could include exploring the role of empirical formulas in chemical manufacturing, environmental monitoring, or pharmaceutical research.
Collaborative Techniques for Teaching Empirical Formulas
Teaching empirical formulas can be a collaborative effort between educators and students. By fostering a culture of shared inquiry and exploration, educators can create an environment conducive to learning and discovery.
- Group discussions and debates
- Peer-to-peer teaching and mentoring
Organize group discussions and debates to stimulate critical thinking and exploration of the empirical formula concept. This encourages students to engage with the material, challenge each other’s understanding, and develop stronger connections to the empirical formula’s significance.
Pair students with peer mentors who have demonstrated a strong grasp of empirical formula concepts. This approach allows students to learn from one another, explore the empirical formula concept in a more personalized manner, and receive targeted support when needed.
“Teaching the empirical formula requires a thoughtful approach that balances hands-on experimentation with theoretical foundations and real-world applications.”
Last Word
In conclusion, finding the empirical formula is a valuable skill that every chemist should possess. By understanding the concept of empirical formulas, we can unlock the secrets of molecular composition and unlock new possibilities in various industries. Whether you’re a student or a seasoned professional, this guide has provided you with the tools and knowledge necessary to find empirical formulas with ease.
FAQ Compilation
What is an empirical formula, and how is it different from a molecular formula?
An empirical formula represents the simplest ratio of atoms in a molecule, whereas a molecular formula represents the actual number of atoms in a molecule.
How do I determine the empirical formula of a compound?
To determine the empirical formula, you need to know the percentage composition of the compound and its molar mass. By dividing the molar mass by the atomic masses of each element, you can obtain the empirical formula.
Can I use the empirical formula to predict the properties of a compound?
Yes, the empirical formula can be used to predict the properties of a compound, such as its melting point, boiling point, and solubility. By understanding the relationship between the empirical formula and the physical properties of a compound, you can make informed predictions about its behavior.