With how to balance equations at the forefront, this topic is essential for any chemistry enthusiast as it helps unlock the secrets behind chemical reactions. Balancing equations can seem like a daunting task, but it’s actually a simple process that requires attention to detail and a basic understanding of chemistry. In this article, we will take a step-by-step approach to guiding you through the process of balancing chemical equations.
From understanding the importance of notation and symbols to applying Hess’s Law and Avogadro’s Law, we will cover it all. Whether you’re a student or a professional, learning how to balance equations is a fundamental skill that will serve you well in the world of chemistry.
Balancing Equations in Chemistry
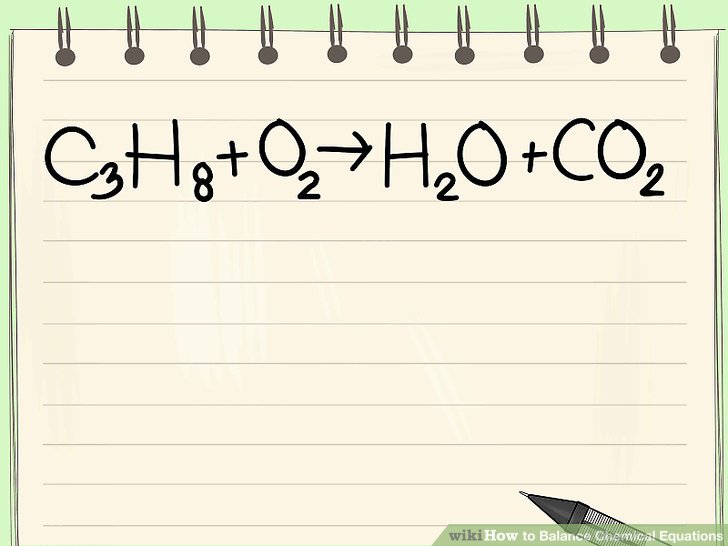
Balancing chemical equations is a crucial concept in chemistry that allows us to understand the stoichiometry of chemical reactions. It is essential for calculating the amount of reactants and products involved in a reaction. A balanced equation is one where the number of atoms of each element is the same on both the reactant and product sides.
Understanding the Process of Balancing Equations
To balance a chemical equation, we need to ensure that the number of atoms of each element is the same on both the reactant and product sides. This is typically done by adding coefficients (numbers that tell us how many molecules of each substance are present) in front of the formulas of the reactants and products. The coefficients can be whole numbers, fractions, or decimals.
| Reactants | Products | Explanation |
|---|---|---|
| Ca + O2 → CaO | To balance the equation, we need 2 oxygen atoms on the reactant side, and we can do this by multiplying O2 by 2. | |
| 2CaO | We now have 2 calcium atoms and 2 oxygen atoms on the product side, which matches the reactant side. |
| Reactants | Products | Explanation |
|---|---|---|
| N2 + O2 → NO2 | To balance the nitrogen atom, we need 2 nitrogen atoms on the product side, so we multiply NO2 by 2. | |
| 2NO2 | However, this introduces an odd number of oxygen atoms on the product side. We can fix this by multiplying O2 by 1.5 (3/2). | |
| However, it’s more common to write the coefficient as 3 to make arithmetic easier. So the balanced equation is: N2 + 3/2O2 → 2NO2. |
| Reactants | Products | Explanation |
|---|---|---|
| CH4 + O2 → CO2 + H2O | To balance the carbon atom, we need 1 carbon atom on the product side, so we multiply CO2 by 1. | |
| To balance the hydrogen atom, we need 4 hydrogen atoms on the product side, so we multiply H2O by 2. | ||
| CO2 + 2H2O | The hydrogen atoms and oxygen atoms on both sides of the equation are now balanced. |
Common Mistakes to Avoid
There are several common mistakes to avoid when balancing chemical equations. Here are a few:
- Mistaking stoichiometric coefficients for empirical formulas.
- Incorrectly assigning the coefficients.
- Using complex algebraic formulas or fractions to write coefficients.
- Writing unbalanced partial equations without a corresponding complete balanced equation.
- Writing unbalanced chemical equations without indicating the reactant or product side.
- Using non-standard, inconsistent, or confusing notation in writing chemical equations.
Case Study: Real-World Importance of Balancing Equations
In chemistry, balancing equations is a crucial concept that is important in various real-world applications. In industrial processes, for example, balancing equations is required for accurate calculations of reactants and products. Balancing equations can help to optimize reactant ratios, minimize waste, and improve efficiency in industrial processes.
Correct Notation and Symbols
Using correct notation and symbols when writing and balancing chemical equations is also crucial. The International Union of Pure and Applied Chemistry (IUPAC) has established specific guidelines for chemical nomenclature and notation, including symbols, punctuation, and capitalization rules. It’s essential to adhere to these guidelines when writing and balancing chemical equations to avoid confusion.
Consequences of Not Balancing Equations
Failing to balance chemical equations can lead to incorrect stoichiometric calculations, which can have serious consequences in various fields. It can lead to:
* Inaccurate product yields and purity
* Increased costs due to incorrect reactant ratios
* Safety risks due to incorrect handling of chemicals
* Inability to scale up or reproduce industrial processes
Conclusion
In conclusion, balancing chemical equations is an essential concept in chemistry that requires careful attention to detail and adherence to specific guidelines. By following these guidelines and avoiding common mistakes, we can ensure accurate calculations, safe handling of chemicals, and optimized industrial processes.
Using Avogadro’s Law to Balance Chemical Equations
Avogadro’s Law provides a fundamental principle in chemistry that governs the behavior of molecules in a gas phase, directly influencing the balancing of chemical equations. By understanding the relationship between the number of molecules and the volume, we can determine the precise mole ratios required for chemical reactions. This connection is vital in ensuring that chemical equations accurately reflect the reactions occurring in nature.
Understanding Avogadro’s Law and Its Application in Chemical Equation Balancing, How to balance equations
Avogadro’s Law, which states that equal volumes of gases at the same temperature and pressure contain an equal number of molecules, provides a basis for understanding the mole ratios in chemical reactions. This fundamental principle is reflected in the ideal gas law, PV = nRT, where P is pressure, V is volume, n is the number of moles, R is the gas constant, and T is temperature. By applying this concept, chemists can confidently balance chemical equations, ensuring that the reactants and products are represented accurately.
Applying Avogadro’s Law to Balance Chemical Equations
Avogadro’s Law is not only limited to ideal gases; it also applies to real-world chemical reactions. By applying the law, chemists can accurately balance chemical equations and predict the behavior of molecules in a gas phase. For instance, consider the following reaction:
| Chemical Equation: | |
|---|---|
| CH₄ + O₂ → CO₂ + H₂O | CH₄ + 2O₂ → CO₂ + 2H₂O |
In the given reaction, Avogadro’s Law dictates that for every molecule of methane (CH₄) reacting with oxygen (O₂), two molecules of oxygen are required to produce one molecule of carbon dioxide (CO₂) and two molecules of water (H₂O).
Relating Avogadro’s Law to the Ideal Gas Law
Avogadro’s Law and the ideal gas law are intimately related, as the law describes the behavior of ideal gases based on their mole ratios. The ideal gas law, PV = nRT, incorporates the concept of mole ratios, allowing chemists to calculate the volume of a gas given the number of moles, pressure, and temperature. For instance, if we know the pressure and volume of a gas at a particular temperature, we can use the ideal gas law to determine the number of moles present.
By analyzing the behavior of gases and applying Avogadro’s Law, we can infer that the ideal gas law is a direct consequence of the law. For example:
| Equation: | Description: |
|---|---|
| PV = nRT | The ideal gas law, where P is pressure, V is volume, n is the number of moles, R is the gas constant, and T is temperature. |
Avogadro’s Law provides a fundamental principle in chemistry, influencing the balancing of chemical equations and the behavior of molecules in a gas phase. By applying this concept, chemists can confidently predict and analyze chemical reactions, ensuring that the reactants and products are represented accurately.
Comparing Avogadro’s Law with Other Methods for Balancing Chemical Equations
Different methods exist for balancing chemical equations, including the use of the oxidation number method, the half-reaction method, and Avogadro’s Law. While Avogadro’s Law focuses on the mole ratios of reactants and products, other methods consider the oxidation numbers and half-reactions involved in the reaction.
| Method | Description |
| :——— | :—————————————————————————————————————————————————————– |
| Avogadro’s Law | Based on the mole ratios of reactants and products, Avogadro’s Law provides a direct approach to balancing chemical equations. |
| Oxidation Number Method | This method involves determining the oxidation numbers of the reactants and products to balance the chemical equation, accounting for electron transfer. |
| Half-Reaction Method | The half-reaction method involves identifying the oxidation and reduction reactions involved in the chemical equation, balancing each half-reaction separately. |
Each method has its advantages and limitations, and the choice of method depends on the specific context of the chemical reaction.
Final Wrap-Up

In conclusion, balancing equations is an essential skill that requires attention to detail and a basic understanding of chemistry. By mastering this process, you will be able to unlock the secrets behind chemical reactions and apply your knowledge to real-world problems.
Detailed FAQs: How To Balance Equations
What is the difference between balancing a chemical equation and balancing a stoichiometric equation?
Balancing a chemical equation involves adjusting the coefficients of the reactants and products to ensure that the law of conservation of mass is satisfied. Balancing a stoichiometric equation, on the other hand, involves ensuring that the number of moles of each reactant and product is in the correct ratio.
Can you use Avogadro’s Law to balance a chemical equation?
Yes, Avogadro’s Law can be used to balance a chemical equation by ensuring that the number of moles of each reactant and product is in the correct ratio. However, it’s essential to use Avogadro’s Law in conjunction with other methods, such as Hess’s Law.
How do I determine the limiting reactant in a chemical reaction?
To determine the limiting reactant, you need to identify which reactant has the smallest mole ratio. The limiting reactant will determine which reactant is in excess. This information is crucial in predicting the products of a chemical reaction.
