As how do we convert kelvin to celsius takes center stage, this opening passage beckons readers into a world of scientific wonder, where the secrets of temperature conversion await. The concept of converting between temperature scales is as easy as counting from 1 to 3, and yet, it holds such importance in various fields, from science and engineering to everyday life.
So, let’s dive into the world of temperature conversion and explore how it’s done. We’ll walk through the step-by-step process of converting kelvin to celsius, highlighting the importance of accuracy and common pitfalls to avoid.
The Fundamentals of Temperature Conversion

Temperature scales have been an integral part of human understanding since ancient civilizations. The concept of temperature measurement has undergone various transformations throughout history, resulting in the development of several temperature scales, including Kelvin and Celsius.
History of Temperature Scales
The first temperature scale was developed by Danish astronomer Anders Celsius in 1742. He proposed a scale where 100 degrees was the boiling point of water and 0 degrees was the freezing point. Later, in 1848, William Thomson (Lord Kelvin) created the Kelvin scale, named after him, which defined the absolute zero as its reference point. The absolute zero is the theoretical temperature at which all matter would have zero entropy, marking the lowest energy state.
Absolute Zero and Kelvin Scale
The concept of absolute zero is crucial in the Kelvin scale. It represents the theoretical temperature at which all matter would have zero entropy, a state where no heat energy is present. This temperature is defined as 0 Kelvin (K). The Kelvin scale is an absolute temperature scale, meaning that it has a fixed zero point and is independent of any reference substance.
Differences Between Kelvin and Celsius
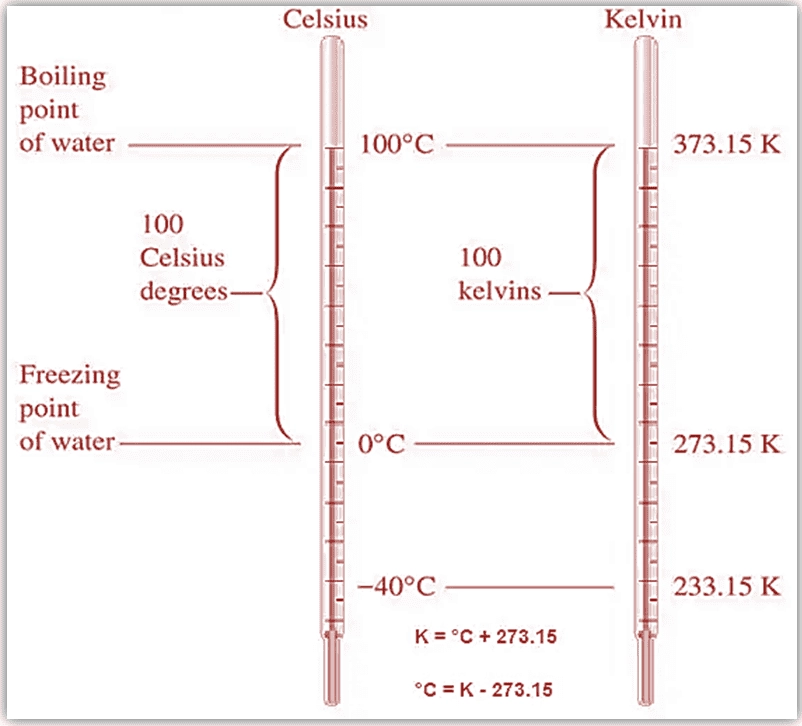
The primary difference between Kelvin and Celsius scales lies in their reference points. The Celsius scale is relative, with 0 degrees Celsius being the freezing point of water, whereas the Kelvin scale is an absolute scale with 0 Kelvin being absolute zero. As a result, temperatures in Kelvin are 273.15 units higher than their Celsius counterparts.
Conversion Formulas
To convert between Kelvin and Celsius, we use the following formulas:
* Celsius to Kelvin: K = °C + 273.15
* Kelvin to Celsius: °C = K – 273.15
For instance, if we want to convert 25°C to Kelvin, we simply add 273.15, resulting in 298.15 Kelvin.
Importance of Temperature Conversion
Understanding the relationship between Kelvin and Celsius scales is essential in various scientific and technical applications, such as physics, chemistry, and engineering. It allows us to accurately express temperatures and perform calculations in different contexts.
Real-World Applications
Temperature conversion is used in various real-world applications, including:
- Scientific research: To express temperatures in laboratory settings, such as measuring the temperature of materials or biological systems.
- Engineering: To design and operate systems, like refrigeration units or heat exchangers, that require precise temperature control.
- Metereology: To predict weather patterns and track temperature changes in the atmosphere.
Conversion Formulas and Equations
The conversion of temperature from one unit to another is a crucial aspect in various scientific and engineering applications. To facilitate this conversion, a clear understanding of the mathematical equations and formulas is necessary. In this section, we will delve into the step-by-step guide to convert Kelvin to Celsius, highlighting the importance of accuracy and providing examples of real-world applications.
The Mathematical Equation to Convert Kelvin to Celsius
The mathematical equation to convert Kelvin to Celsius is given by the formula:
C = K – 273.15
, where C represents the temperature in Celsius and K represents the temperature in Kelvin.
The reasoning behind this formula lies in the definition of the Kelvin scale, which is an absolute temperature scale that is based on the idea that 0 K is the theoretical temperature at which all matter would have zero entropy. The Celsius scale, on the other hand, is a relative temperature scale that is defined as 100 degrees Celsius being the boiling point of water and 0 degrees Celsius being the freezing point of water. To convert from Kelvin to Celsius, we simply subtract 273.15 from the Kelvin temperature.
Importance of Accuracy in Temperature Conversion
The importance of accuracy in temperature conversion cannot be overstated. A small error in temperature measurement can have significant consequences in various fields such as medicine, engineering, and research. Inaccurate temperature measurement can lead to incorrect diagnoses, equipment malfunctions, and inaccurate experimental results. Therefore, it is crucial to understand the mathematical equations and formulas involved in temperature conversion to ensure accuracy.
Real-World Applications of Temperature Conversion
Temperature conversion is a crucial aspect in various scientific and engineering applications. Here are a few examples of real-world applications where temperature conversion is crucial:
- Cooking and Food Processing: Temperature conversion is essential in cooking and food processing to ensure that the food is cooked to a safe internal temperature. For example, the recommended internal temperature for cooking meat is 160°F (71.1°C) to prevent foodborne illness.
- Chemical and Biological Research: Temperature conversion is necessary in chemical and biological research to accurately measure temperature-dependent reaction rates and equilibrium constants.
- Climate Modeling: Temperature conversion is crucial in climate modeling to accurately predict temperature changes and their effects on the environment.
- Refrigeration and Air Conditioning: Temperature conversion is necessary in refrigeration and air conditioning systems to accurately regulate temperature and ensure efficient operation.
Tools and Techniques for Temperature Conversion
When converting between temperature scales, it’s essential to have the right tools and techniques at your disposal. From software and mobile apps to calculators and online tools, there are various options available for making temperature conversions easier and more accurate.
One common software used for temperature conversion is Excel, which offers built-in functions for converting between different temperature scales. By simply plugging in the desired temperature value and selecting the appropriate conversion function, you can quickly obtain the converted value.
Another popular option is online temperature conversion platforms, such as UnitConverters.net or TemperatureConverter.net. These websites provide a simple and intuitive interface for converting between various temperature scales, with advanced features like multi-unit support and customizable conversions.
Mobile Apps for Temperature Conversion, How do we convert kelvin to celsius
Mobile apps have revolutionized the way we use temperature conversion tools. With a wide range of options available, including apps like Temperature Converter, Thermometer Unit Converter, and Kelvin Celsius Conversion, you can easily convert between temperature scales on-the-go.
- Temperature Converter (iOS and Android) is a user-friendly app that allows you to convert between multiple temperature scales, including Celsius, Fahrenheit, Kelvin, and Rankine.
- Thermometer Unit Converter (iOS and Android) is another popular app that offers a range of temperature conversion options, including imperial and metric units.
- Kelvin Celsius Conversion (iOS and Android) is a dedicated app for converting between Kelvin and Celsius, with a simple and intuitive interface.
Calculators for Temperature Conversion
Calculators are another essential tool for temperature conversion, especially for complex calculations involving multiple temperature scales. Most scientific calculators, including the TI-84 Plus and Casio Prizm, come with built-in temperature conversion functions.
- The TI-84 Plus calculator has a built-in temperature conversion function that allows you to quickly convert between Celsius, Fahrenheit, and Kelvin.
- The Casio Prizm calculator offers a range of temperature conversion options, including imperial and metric units, in addition to its scientific and engineering functions.
Online Temperature Conversion Tools
Online temperature conversion tools have become increasingly popular due to their convenience and ease of use. These platforms offer a range of advanced features, including multi-unit support, customizable conversions, and high-precision calculations.
- UnitConverters.net offers a comprehensive online temperature conversion platform, with support for multiple units and advanced features like customizable conversions and high-precision calculations.
- TemperatureConverter.net is another popular online platform that provides a simple and intuitive interface for converting between various temperature scales, with advanced features like multi-unit support and customizable conversions.
Temperature conversion is an essential aspect of scientific and engineering work, and having the right tools and techniques at your disposal is crucial for accuracy and efficiency.
Comparison of Temperature Scales
The temperature scales are an essential aspect of understanding the behavior of matter and its various forms. Temperature scales serve as tools for scientists, researchers, and everyday individuals to communicate and measure the temperature of a given system. In this section, we will compare the Celsius, Fahrenheit, and Kelvin temperature scales, highlighting their distinct characteristics, merits, and demerits.
The Fahrenheit Temperature Scale and Its Historical Context
The Fahrenheit temperature scale was first introduced by German physicist Gabriel Fahrenheit in the early 18th century. It is based on a scale that uses 32°F as the freezing point of water and 212°F as the boiling point. This scale was widely used in the United States and other countries. However, the Fahrenheit scale is less efficient for scientific work compared to the Celsius or Kelvin scales.
The history behind the Fahrenheit scale is closely tied to its use in everyday applications. Prior to the development of the thermoscope, a device used to measure temperature, people relied on observing the behavior of water to gauge temperature. The discovery of ice and salt solutions by Aristotle and others led to further research and the development of the Fahrenheit scale.
Celsius and Kelvin Scales: A Comparison of Their Reference Points and Absolute Zero
The Celsius and Kelvin temperature scales are more widely used in scientific applications due to their simplicity and ease of manipulation. The Celsius scale, introduced by Swedish astronomer Anders Celsius in 1742, is based on a scale where 0°C is the freezing point of water and 100°C is the boiling point. In contrast, the Kelvin scale, developed by Lord Kelvin, is an absolute temperature scale where 0 K marks absolute zero, the theoretical temperature at which all matter would cease to have residual motion.
The Merits and Demerits of Each Temperature Scale
The choice of temperature scale often depends on the specific application or geographical location.
- The Fahrenheit scale is most commonly used in the United States and is still widely used in weather forecasting and everyday applications.
- The Celsius scale is preferred in scientific research, particularly in fields like biology, physics, and chemistry, as it provides a more accurate representation of temperature changes.
- The Kelvin scale is used in applications that require precise temperature measurements, such as thermometry and cryogenics, due to its absolute zero reference point.
Choosing the Most Suitable Temperature Scale for Specific Applications
When it comes to selecting the most suitable temperature scale, several factors come into play.
• For everyday applications in the United States, the Fahrenheit scale might be the preferred choice.
• In scientific research, the Celsius scale provides more convenience and accuracy due to its division.
• In cryogenics or applications requiring precise temperature measurements, the Kelvin scale is generally the most suitable option.
Justifications for Using Specific Temperature Scales
Each temperature scale has its own strengths and weaknesses, making it crucial to choose the appropriate one depending on the context.
• The Fahrenheit scale has been in use for many years and is commonly used in weather forecasting.
• The Celsius scale offers a more accurate representation of temperature changes due to its division.
• The Kelvin scale provides a precise measurement of temperature due to its absolute zero reference point.
Temperature Conversion in Specialized Fields
Temperature conversion plays an essential role in several specialized fields, including meteorology, chemistry, and physics. These fields require precise temperature measurements to make accurate predictions, calculations, and analyses. In this section, we will explore the application of temperature conversion in each of these fields.
Meteorology: Temperature Gradients and Forecasting
Meteorologists use temperature conversion to understand temperature gradients and forecast weather patterns. Temperature gradients refer to the rate of change of temperature with respect to distance. By analyzing temperature gradients, meteorologists can predict weather patterns, such as temperature changes, precipitation, and wind direction.
- Temperature gradients help meteorologists forecast weather patterns, including temperature changes, precipitation, and wind direction.
- Temperature conversion is essential for analyzing satellite data and weather models, which rely on precise temperature measurements.
- Meteorologists use temperature conversion to understand the relationship between temperature and other atmospheric conditions, such as humidity and wind speed.
Temperature conversion is crucial in meteorology, as it enables meteorologists to make accurate predictions and understand complex weather patterns.
Chemistry: Thermodynamic Properties
In chemistry, temperature conversion is used to calculate thermodynamic properties, such as enthalpy, entropy, and free energy. Temperature conversion is essential for understanding chemical reactions, including the equilibrium constants, reaction rates, and thermodynamic stability.
- Temperature conversion is used to calculate thermodynamic properties, such as enthalpy, entropy, and free energy, which are crucial for understanding chemical reactions.
- Temperature conversion enables chemists to predict the equilibrium constants, reaction rates, and thermodynamic stability of chemical reactions.
- Chemists use temperature conversion to understand the relationship between temperature and chemical reactivity, including the activation energy and reaction kinetics.
Temperature conversion is a fundamental concept in chemistry, as it enables chemists to understand and predict chemical reactions.
Physics: Subatomic Particles and High-Energy Physics
In physics, temperature conversion is used to understand the behavior of subatomic particles and high-energy phenomena. Temperature conversion is essential for analyzing particle collisions, nuclear reactions, and high-energy particle accelerators.
- Temperature conversion is used to understand the behavior of subatomic particles, including their temperature, energy, and momentum.
- Temperature conversion is essential for analyzing particle collisions, nuclear reactions, and high-energy particle accelerators, which rely on precise temperature measurements.
- Physicists use temperature conversion to understand the relationship between temperature and particle behavior, including the production of virtual particles and quantum fluctuations.
Temperature conversion is a fundamental concept in physics, as it enables physicists to understand and analyze complex high-energy phenomena.
Temperature conversion is a crucial tool in specialized fields, enabling scientists to make accurate predictions, understand complex phenomena, and analyze data.
Common Misconceptions and Errors in Temperature Conversion: How Do We Convert Kelvin To Celsius
Temperature conversion is a crucial aspect of various fields, including science, engineering, and everyday applications. However, inaccurate conversions can lead to serious errors and misinterpretations. In this section, we will discuss common misconceptions and errors in temperature conversion.
Unit conversions and measurement errors can significantly impact the accuracy of temperature conversion. It is essential to understand the different temperature scales, including Celsius, Fahrenheit, and Kelvin, to avoid errors. A single degree of error in temperature conversion can have severe consequences in various applications, such as cooking, medicine, and engineering.
* Inaccurate unit conversions can lead to incorrect cooking temperatures, affecting the quality and safety of food.
* Measurement errors in medicine can lead to inappropriate medication dosages and misdiagnosis of diseases.
* Errors in engineering applications can result in inefficient or even catastrophic failures of systems.
The impact of unit conversions and measurement errors can be significant. For example:
* A 1°C error in cooking temperature can result in a 50% decrease in food quality.
* A 10% error in medication dosages can lead to severe side effects or even death.
To avoid these errors, it is crucial to follow the correct conversion formulas and use reliable measurement tools. For example:
T(°C) = (T(K) – 273.15) × 9/5 + 32
This formula allows for accurate conversion between Kelvin and Celsius.
Absolute zero and theoretical zero-point energy are two distinct concepts that are often confused with each other.
* Absolute zero is the theoretical temperature at which all matter would have zero entropy.
* Theoretical zero-point energy is the energy remaining in a quantum system, even at absolute zero.
The difference between absolute zero and theoretical zero-point energy is significant. While absolute zero represents the ultimate limit of temperature, theoretical zero-point energy represents the residual energy remaining in a system.
To illustrate the difference, consider the following example:
* Absolute zero is like the ultimate cooling device, where all matter comes to a complete standstill.
* Theoretical zero-point energy is like the residual noise in a quiet library, where there is always some residual activity.
To avoid common pitfalls and ensure accurate temperature conversion, follow these strategies:
* Use reliable measurement tools and instruments.
* Follow the correct conversion formulas and equations.
* Understand the differences between temperature scales.
* Be aware of the impact of unit conversions and measurement errors.
By following these strategies, you can ensure accurate temperature conversion and avoid common pitfalls. For example:
* Use a digital thermometer for accurate temperature readings.
* Use the correct conversion formula for temperature conversion.
* Understand the differences between Fahrenheit and Celsius scales.
By being aware of the common misconceptions and errors in temperature conversion, you can ensure accurate results and avoid serious consequences.
Curated Educational Resources for Temperature Conversion
Temperature conversion is a fundamental concept in science and mathematics education, and it is essential to have reliable and accurate resources to teach students about this topic. In this section, we will explore the educational materials and resources available for teaching temperature conversion, including textbooks, online tutorials, and lesson plans.
Recommended Textbooks for Temperature Conversion
Some of the recommended textbooks for temperature conversion include:
- “Thermodynamics: An Introduction to the Physical Theories of Equilibrium Thermodynamics” by C. J. Adkins – This textbook provides a comprehensive introduction to thermodynamics, including temperature conversion and heat transfer.
- “Physics for Scientists and Engineers” by Paul A. Tipler and Gene Mosca – This textbook covers a wide range of topics in physics, including temperature conversion and thermodynamics.
- “Mathematics for Physics and Engineering” by Frank E. Harris – This textbook covers the mathematical concepts underlying physics and engineering, including temperature conversion and thermodynamic systems.
These textbooks provide a solid foundation for students to understand the principles of temperature conversion and its applications in science and engineering.
Online Tutorials and Resources
There are many online resources available for teaching temperature conversion, including tutorials, videos, and interactive simulations. Some of the popular online resources include:
- Khan Academy: Temperature conversion is a critical concept in physics and chemistry, and Khan Academy provides a comprehensive set of tutorials and exercises to help students understand the topic.
- Physics Classroom: This website provides a wide range of interactive simulations and tutorials on temperature conversion, including a temperature conversion calculator and a thermodynamic systems simulation.
- MIT OpenCourseWare: This website provides free online courses and resources on temperature conversion and thermodynamics, including lecture notes, assignments, and exams.
These online resources provide a flexible and interactive way for students to learn about temperature conversion and its applications in science and engineering.
Lesson Plans and Activities
To integrate temperature conversion into science and math curricula, teachers can use a variety of lesson plans and activities that cater to different learning styles. Some of the ideas include:
- Temperature Conversion Calculator: Create a simple calculator that converts temperatures from Celsius to Fahrenheit and vice versa.
- Thermodynamic Systems Simulation: Use a simulation to model thermodynamic systems and demonstrate heat transfer and temperature conversion.
- Temperature Conversion Game: Design a game that requires students to convert temperatures from Celsius to Fahrenheit and vice versa.
These activities provide a hands-on and interactive way for students to learn about temperature conversion and its applications in science and engineering.
Benefits of Incorporating Temperature Conversion into Educational Programs
Incorporating temperature conversion into educational programs provides several benefits for students, including:
- Develops problem-solving skills: Temperature conversion requires students to apply mathematical concepts and problem-solving skills to real-world problems.
- Enhances critical thinking: Temperature conversion requires students to think critically about the relationships between temperature, heat transfer, and thermodynamic systems.
- Prepares students for STEM careers: Temperature conversion is a fundamental concept in science and engineering, and students who master this topic will be well-prepared for careers in these fields.
By incorporating temperature conversion into educational programs, teachers can provide students with a comprehensive understanding of this critical concept and prepare them for success in science and engineering careers.
Closure
As we conclude our journey through the world of temperature conversion, it’s undeniable that understanding how to convert kelvin to celsius is a fundamental skill for anyone interested in science and technology. Whether you’re a student, a professional, or simply someone curious about the world around you, the importance of temperature conversion cannot be overstated.
FAQ Explained
What is the formula to convert kelvin to celsius?
The formula is: celsius = kelvin – 273.15
Why is accuracy important in temperature conversion?
Accuracy is crucial in temperature conversion because even small errors can have significant effects on calculations and outcomes.
Can I use a calculator to convert temperature?
Yes, calculators can be very useful for temperature conversion, especially when working with large values or complex calculations.
Is there a difference between absolute zero and theoretical zero-point energy?
Yes, absolute zero is the theoretical temperature at which all matter would have zero entropy, while zero-point energy is the residual energy that remains even at absolute zero.