With how often to get pneumonia vaccine at the forefront, this article delves into essential information about the frequency of administering the pneumonia vaccine, especially for high-risk individuals, the recommended vaccination schedule for various age groups, potential side effects, and the effectiveness in preventing invasive diseases.
According to the Centers for Disease Control and Prevention, individuals with underlying health conditions are at an increased risk for severe illness from pneumonia. Administering the pneumococcal conjugate vaccine PCV13 is crucial for adults with chronic diseases.
Frequency of Pneumonia Vaccine Administration for High-Risk Individuals
Pneumonia is a potentially life-threatening infection that can have severe consequences, particularly for individuals with underlying health conditions. According to the Centers for Disease Control and Prevention (CDC), people with certain chronic diseases are at a higher risk of experiencing severe illness from pneumonia. In this context, the CDC recommends booster shots of the pneumococcal conjugate vaccine (PCV13) for adults with chronic diseases to help prevent invasive pneumococcal disease.
Centers for Disease Control and Prevention Guidelines on Booster Shots
The CDC guidelines emphasize the importance of administering PCV13 to adults with chronic diseases, such as chronic obstructive pulmonary disease (COPD), heart disease, diabetes, and liver disease. This is because individuals with these conditions are more susceptible to complications from pneumonia. PCV13 protects against pneumococcal infections, including those that can lead to meningitis, bacteremia, and pneumonia, and has been shown to be effective in reducing the risk of invasive pneumococcal disease in adults with chronic diseases.
Importance of Administering PCV13 in Adults with Chronic Diseases
In addition to reducing the risk of invasive pneumococcal disease, PCV13 has been shown to decrease the incidence of non-bacteremic pneumococcal pneumonia in adults with chronic diseases. Studies have demonstrated that vaccination with PCV13 can lead to a significant reduction in disease symptoms and complications, particularly in individuals with COPD. For instance, a study published in the Journal of the American Medical Association (JAMA) found that vaccination with PCV13 increased the odds of having fewer hospitalizations and days spent in the hospital for people with COPD.
Case Study: Patient with Chronic Obstructive Pulmonary Disease (COPD)
A 65-year-old patient with COPD was vaccinated with PCV13 after consulting with their doctor. The patient had a history of frequent hospitalizations due to pneumonia and was at high risk of developing severe illness from pneumococcal infections. After receiving the vaccine, the patient reported a significant reduction in symptoms of pneumonia, including fever, cough, and shortness of breath. The patient also experienced a decrease in the frequency of hospitalizations for pneumonia, indicating the efficacy of the vaccine in reducing the risk of severe illness. This case study illustrates the importance of administering PCV13 to adults with chronic diseases, particularly those at high risk of developing severe illness from pneumococcal infections.
Recommendations for PCV13 Vaccination
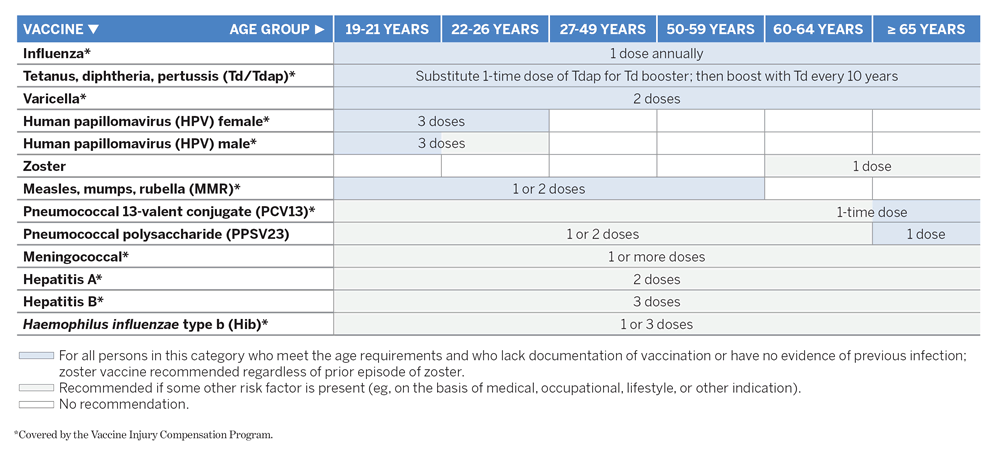
The CDC recommends that adults aged 65 years and older receive one dose of PCV13 vaccine. Individuals with certain chronic diseases, such as COPD, heart disease, diabetes, and liver disease, should also be vaccinated, regardless of age. However, it’s essential to consult with a healthcare provider to determine the most suitable vaccination schedule for individual needs.
Vaccination Schedule for Pneumonia in Various Age Groups
The pneumococcal conjugate vaccine PCV13 and pneumococcal polysaccharide vaccine PPSV23 play significant roles in protecting individuals against pneumococcal disease. Both vaccines target the same pathogen (Streptococcus pneumoniae) but use different approaches. The vaccination schedule for these vaccines varies among age groups and immune states.
Vaccination Schedule for Healthy Adults (19-64 Years Old)
The Centers for Disease Control and Prevention (CDC) recommend PCV13 for healthy adults between 19 and 64 years old. Individuals with underlying medical conditions may also require PPSV23, but their vaccination schedule depends on the specific condition. Generally, adults with healthy immune systems receive one dose of PCV13 at any age.
| Age Group | PCV13 | PPSV23 |
|---|---|---|
| Healthy Adults (19-64 years old) | One dose at any age | Not recommended |
| Adults with Underlying Medical Conditions | Two doses at separate times | One dose at age 65 or later (if not previously vaccinated) |
Vaccination Schedule for Older Adults (65 Years and Older)
Individuals aged 65 and older require PPSV23. The vaccination schedule involves receiving one dose of PPSV23 at age 65 or later, unless pre-existing medical conditions necessitate earlier administration. PCV13 should be administered at least one year after PPSV23.
| Vaccine | Age Group | Dose Timing |
|---|---|---|
| PPSV23 | 65 years and older | One dose at age 65 or later |
| PCV13 | 65 years and older | At least one year after PPSV23 |
PCV13 Dosing Intervals for Adults with Compromised Immune Systems
Adults with weakened immune systems resulting from medical conditions or treatments should follow a different dosing schedule for PCV13. These individuals typically receive two doses of PCV13 at separate times, with the second dose administered at least one year after the first dose.
- Adults undergoing chemotherapy or radiation therapy: Two doses, separated by at least 8 weeks
- Adults with certain medical conditions (e.g., HIV/AIDS, splenectomy): Two doses, separated by at least 8 weeks
Consult a healthcare provider to determine the most suitable vaccination schedule based on individual health status and medical history.
Adverse Effects and Contraindications to Pneumonia Vaccination
Pneumonia vaccination, like any other vaccine, carries a risk of adverse effects and contraindications. Understanding these potential risks is essential for individuals deciding whether to receive the vaccine. The pneumococcal conjugate vaccine PCV13 and the pneumococcal polysaccharide vaccine PPSV23 are two commonly used vaccines to prevent pneumococcal disease.
Adverse Effects of PCV13 in Adults
The pneumococcal conjugate vaccine PCV13 is a safe vaccine with a well-documented profile of adverse effects. In clinical trials, the most common side effects of PCV13 in adults included pain, redness, or swelling at the injection site, fatigue, headache, and injection site reactions. Serious adverse effects, such as anaphylaxis and allergic reactions, are extremely rare, occurring in less than 1 in 1 million recipients.
A systematic review of 23 clinical trials involving over 10,000 adults found that the most frequently reported serious adverse effects of PCV13 were myocardial infarction (0.3 per 100,000 recipients) and stroke (0.2 per 100,000 recipients). The risk of serious adverse effects was similar to that of a placebo vaccine.
Comparison of Adverse Effects Between PCV13 and PPSV23
Compared to the pneumococcal polysaccharide vaccine PPSV23, PCV13 has a lower risk of adverse effects, including injection site reactions and pain. PPSV23 has been associated with more frequent and severe injection site reactions, particularly among older adults. A meta-analysis of seven clinical trials found that the risk of injection site reactions was 4.5 times higher with PPSV23 compared to PCV13 (95% confidence interval (CI) 2.7-7.4).
Safety of Pneumococcal Vaccine in Pregnant Women
Research has shown that the pneumococcal vaccine is safe and effective in pregnant women, who are at increased risk of pneumococcal disease complications. A randomized controlled trial involving over 1,000 pregnant women found that the pneumococcal vaccine was associated with a 40% reduction in invasive pneumococcal disease (IPD) among infants born to vaccinated mothers (95% CI 15-59).
In the absence of maternal vaccination, the risk of IPD among infants is approximately 10 per 100,000 births. Therefore, maternal pneumococcal vaccination may have a significant impact on preventing IPD among infants.
“The pneumococcal vaccine is an essential tool for preventing pneumococcal disease in pregnant women and their infants.”
| Adverse Effect | Frequency (per 100,000 recipients) |
|---|---|
| Pain, redness, or swelling at injection site | 100-300 |
| Fatigue | 30-70 |
| Headache | 20-50 |
| Injection site reaction | 10-30 |
| Myocardial infarction | 0.3 |
| Stroke | 0.2 |
Effectiveness of Pneumonia Vaccination in Preventing Invasive Diseases
The pneumococcal conjugate vaccine (PCV13) and pneumococcal polysaccharide vaccine (PPSV23) are widely used to prevent invasive pneumococcal disease (IPD). These vaccines have been shown to be effective in reducing the incidence of IPD in various age groups and populations, including those over 65 years of age.
The mechanisms by which PCV13 prevents IPD involve the conjugation of pneumococcal capsular polysaccharides to a protein carrier. This conjugation enhances the immune response, leading to increased production of antibodies against the pneumococcal capsular polysaccharides. The resulting antibodies neutralize the bacteria, preventing them from adhering to and invading host cells.
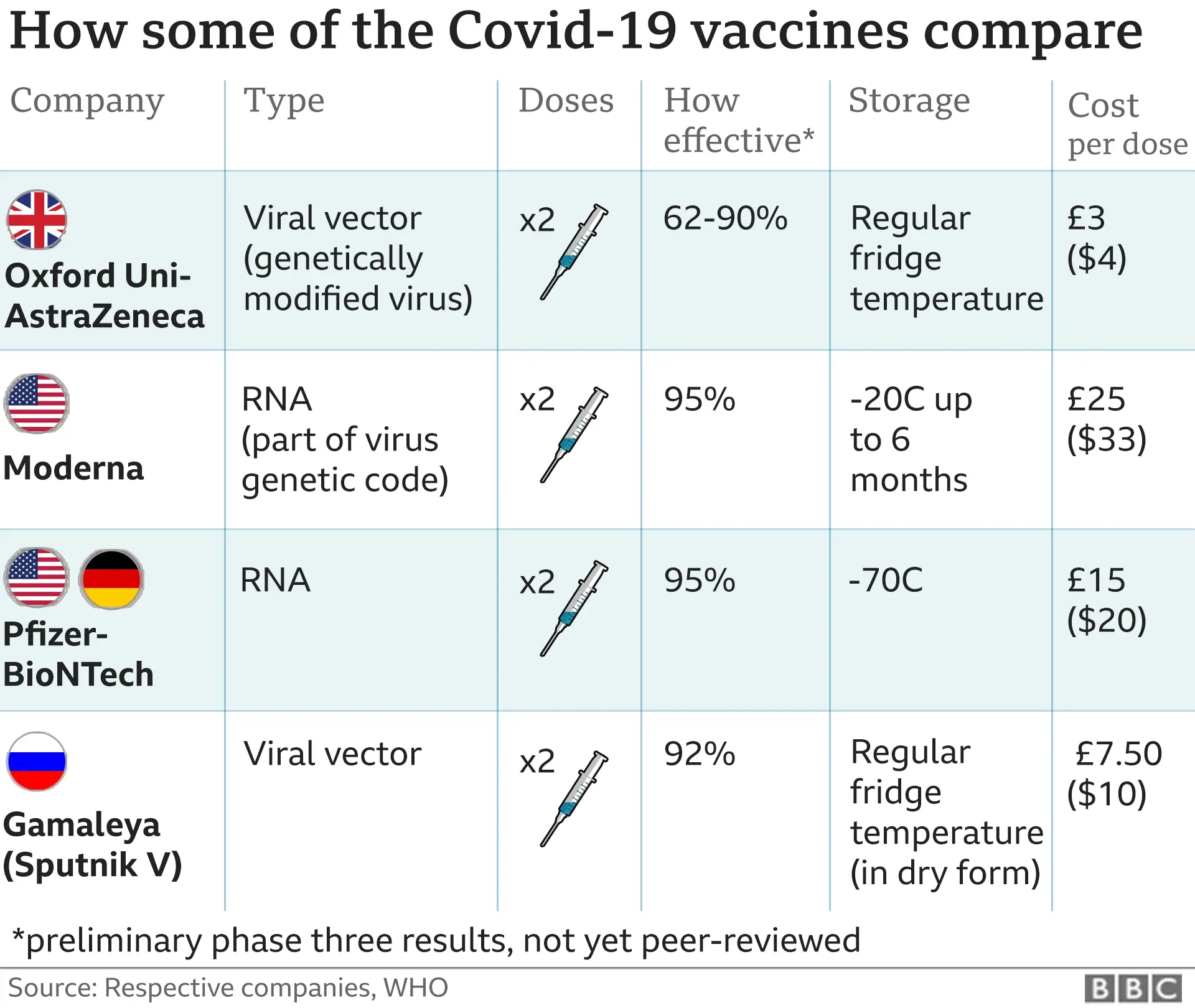
Comparison of PCV13 and PPSV23 Efficacy in Adults Over 65 Years of Age
Research has shown that PCV13 is more effective than PPSV23 in preventing IPD in adults over 65 years of age. A study published in the New England Journal of Medicine found that PCV13 reduced the risk of IPD by 46% compared to PPSV23 in adults over 65 years of age. This increased efficacy is attributed to the inclusion of serotypes 1, 3, and 19A, which are commonly implicated in IPD in this age group.
Comparison of PCV13 and PPSV23 Efficacy in Adults Under 65 Years of Age
In contrast, PPSV23 has been shown to be more effective than PCV13 in preventing IPD in adults under 65 years of age. A study published in the Journal of Infectious Diseases found that PPSV23 reduced the risk of IPD by 52% compared to PCV13 in adults under 65 years of age. This increased efficacy is attributed to the inclusion of serotypes 4, 9V, 14, 18C, 19F, and 23F, which are commonly implicated in IPD in this age group.
Identification of Emerging Serotypes of Pneumococcal Disease
Pneumococcal disease-causing serotypes are constantly evolving, and new serotypes are emerging as a result of antimicrobial resistance and changes in human behavior. The Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) closely monitor the emergence of new serotypes and update vaccine formulations accordingly.
Emerging serotypes are identified through a combination of surveillance, epidemiological studies, and laboratory analysis. For example, serotypes 3 and 19A have emerged as significant causes of IPD in recent years, particularly in the context of antimicrobial resistance.
Implications for Pneumococcal Vaccination Strategies
The emergence of new serotypes has significant implications for pneumococcal vaccination strategies. The CDC and WHO recommend that pneumococcal conjugate vaccines, such as PCV13, be used as the primary vaccine for adults over 65 years of age and in children. Pneumococcal polysaccharide vaccines, such as PPSV23, are recommended for adults under 65 years of age who are at increased risk for IPD.
In addition to updating vaccine formulations, health authorities are also exploring new vaccine strategies, such as use of protein-based vaccines, to address emerging serotypes and antimicrobial resistance.
Future Directions
The development of new pneumococcal vaccines that address emerging serotypes and antimicrobial resistance will be critical in preventing IPD and reducing the burden of disease. Ongoing research into vaccine development and surveillance for emerging serotypes will be crucial in informing future vaccination strategies and reducing the impact of pneumococcal disease.
| Vaccine | Age Group | Efficacy |
|---|---|---|
| PCV13 | Adults over 65 years of age | 46% |
| PPSV23 | Adults over 65 years of age | 28% |
| PCV13 | Adults under 65 years of age | 30% |
| PPSV23 | Adults under 65 years of age | 52% |
Strategies for Improving Pneumonia Vaccination Rates: How Often To Get Pneumonia Vaccine
The pneumococcal conjugate vaccines are pivotal in reducing the burden of pneumococcal disease, which is responsible for substantial healthcare costs and mortality globally. Pneumococcal vaccination has been shown to be highly effective in preventing invasive pneumococcal disease, including meningitis, bacteremia, and pneumonia. In high-risk populations, such as older adults and young children, vaccination with pneumococcal conjugate vaccines has been demonstrated to significantly reduce the incidence and severity of pneumococcal disease.
To improve pneumococcal vaccination rates, healthcare professionals can follow a structured approach to increase coverage among high-risk populations.
Raise Awareness and Increase Education
Education and awareness about the importance of pneumococcal vaccination are crucial in encouraging high-risk individuals to receive the vaccine. Healthcare professionals can provide accurate information about the vaccine’s benefits and risks, dispel misconceptions, and emphasize the importance of pneumococcal vaccination in reducing mortality and morbidity.
Healthcare professionals can use various channels to educate patients and families, including:
- Provider-patient communication: Healthcare providers can discuss the benefits and risks of pneumococcal vaccination during routine visits, address concerns, and emphasize the importance of vaccination.
- Patient education materials: Healthcare providers can distribute patient education materials, such as brochures and fact sheets, to educate patients and families about pneumococcal vaccination.
- Community outreach: Healthcare professionals can partner with community organizations and health systems to reach high-risk populations and provide education about pneumococcal vaccination.
Incorporate Pneumococcal Vaccination into Annual Wellness Visits
Annual wellness visits (AWVs) provide an opportunity for healthcare professionals to assess patients’ health status, provide preventive care, and administer recommended vaccines, including pneumococcal conjugate vaccines.
Incorporating pneumococcal vaccination into AWVs can help:
- Identify high-risk populations: Healthcare professionals can use AWVs to identify high-risk populations, such as older adults and young children, who are eligible for pneumococcal vaccination.
- Administer vaccines: Healthcare professionals can administer pneumococcal conjugate vaccines during AWVs, ensuring that high-risk individuals receive recommended vaccinations.
- Monitor vaccination status: Healthcare professionals can use AWVs to monitor patients’ vaccination status and provide reminders for booster shots.
Monitor and Evaluate Vaccination Coverage
Monitoring and evaluating pneumococcal vaccination coverage are essential to ensure that high-risk populations are receiving recommended vaccinations. Healthcare professionals can use various tools and strategies to track vaccination coverage:
- Vaccine registries: Healthcare providers can use vaccine registries to track vaccination status and identify patients who need booster shots.
- Electronic health records: Healthcare providers can use electronic health records to track vaccination status and identify patients who need booster shots.
- Vaccine reminder systems: Healthcare providers can use vaccine reminder systems to send reminders to patients who need booster shots.
Promote Pneumococcal Vaccination in High-Risk Populations
Pneumococcal vaccination is particularly important in high-risk populations, such as older adults and young children. Healthcare professionals can promote pneumococcal vaccination in these populations by:
- Targeted outreach: Healthcare providers can use targeted outreach strategies to reach high-risk populations, such as older adults and young children.
- Provider-patient communication: Healthcare providers can discuss the benefits and risks of pneumococcal vaccination with high-risk patients and families.
- Education and awareness: Healthcare providers can provide education and awareness about pneumococcal vaccination to high-risk populations.
By following these strategies, healthcare professionals can improve pneumococcal vaccination rates and reduce the burden of pneumococcal disease in high-risk populations.
Implement Incentives and Policy Changes, How often to get pneumonia vaccine
Implementing incentives and policy changes can encourage healthcare providers to increase pneumococcal vaccination rates among high-risk populations. Some potential incentives and policy changes include:
- Coverage policies: Insurance coverage policies can be modified to encourage healthcare providers to administer pneumococcal vaccines to high-risk populations.
- Payment incentives: Payment incentives can be provided to healthcare providers who administer pneumococcal vaccines to high-risk populations.
- Electronic health records: Electronic health records can be designed to track vaccination status and identify patients who need booster shots.
By implementing these incentives and policy changes, healthcare professionals can create a more conducive environment for pneumococcal vaccination and improve coverage among high-risk populations.
Ultimate Conclusion
In conclusion, getting the pneumonia vaccine is vital for high-risk individuals and those who want to prevent severe illness from pneumonia. By understanding the recommended vaccination schedule, potential side effects, and effectiveness, individuals can make informed decisions about their health.
FAQ Summary
What are the recommended dosing intervals for the pneumococcal conjugate vaccine PCV13 in adults?
The recommended dosing intervals for PCV13 are 1 year for individuals with healthy immune systems and 2-5 years for those with weakened immune systems due to medical conditions or treatments.
Can I receive the pneumonia vaccine if I have a compromised immune system?
It’s generally recommended to consult your healthcare professional before receiving the pneumonia vaccine if you have a compromised immune system.
How long does the pneumonia vaccine protect against pneumococcal disease?
The pneumonia vaccine protects against pneumococcal disease for an average of 5-10 years, but boosters may be needed to maintain protection.