As how to calculate ph takes center stage, this comprehensive guide provides readers with an in-depth knowledge of the fundamental principles, different methods, and common tools used in pH level measurements. Understanding the importance of pH calculations in various fields, including environmental and industrial applications, is crucial for accurate and reliable results.
The following Artikel covers the essential topics and discussions related to pH calculations, including the understanding of pH measurements, choosing the right pH calculation method, calculating pH at different concentrations, and more.
Choosing the Right pH Calculation Method
pH calculation methods are fundamental in various scientific and industrial applications, such as chemistry, biology, and environmental science. Each method offers a unique approach to understanding pH values, and selecting the most suitable method is crucial to ensure accurate results.
The Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation is a widely used method for calculating pH values. It is based on the equilibrium pH equation, which relates the concentrations of the acidic and basic components of a solution.
\[ \textpH = \textpK_a + \log \frac[\textA^-][\textHA] \]
This equation is particularly useful for calculating the pH of buffer solutions, which are mixtures of a weak acid and its conjugate base. The Henderson-Hasselbalch equation is a simplification of the more complex equilibrium pH equation, and it is often used as a shortcut to quickly estimate pH values.
The Sorensen Method, How to calculate ph
The Sorensen method is another pH calculation method that is based on the measurement of the concentration of hydrogen ions in a solution. This method involves the use of a pH meter, which measures the potential difference between two electrode terminals immersed in the solution. The Sorensen method is often used in situations where the Henderson-Hasselbalch equation cannot be applied, such as in the calculation of the pH of very strong or very weak acids.
Determining the Appropriate Method
When deciding which pH calculation method to use, several factors must be considered. The type of solution being analyzed, the accuracy required, and the available equipment and resources are all important considerations. In general, the Henderson-Hasselbalch equation is suitable for buffer solutions and other mixtures of weak acids and their conjugate bases, while the Sorensen method is suitable for situations where the Henderson-Hasselbalch equation cannot be applied.
- Accurate pH measurement: Choose the Sorensen method when high accuracy is required, such as in quality control or research applications.
- Buffer solutions: Use the Henderson-Hasselbalch equation to calculate the pH of buffer solutions, which are mixtures of a weak acid and its conjugate base.
- Strong or weak acids: Choose the Sorensen method when dealing with very strong or very weak acids, where the Henderson-Hasselbalch equation is not applicable.
In conclusion, choosing the right pH calculation method depends on the specific requirements of the task. By considering the type of solution being analyzed, the accuracy required, and the available equipment and resources, researchers and scientists can select the most suitable method for their needs.
Calculating pH at Different Concentrations: How To Calculate Ph
pH calculations are crucial in various applications, including chemistry experiments, quality control in industries, and environmental monitoring. Accurate pH measurements can help ensure the safety and efficacy of substances, as well as prevent contamination and damage to equipment. In this guide, we will discuss how to calculate pH at different concentrations of strong and weak acids and bases using various methods.
Strong Acid pH Calculations
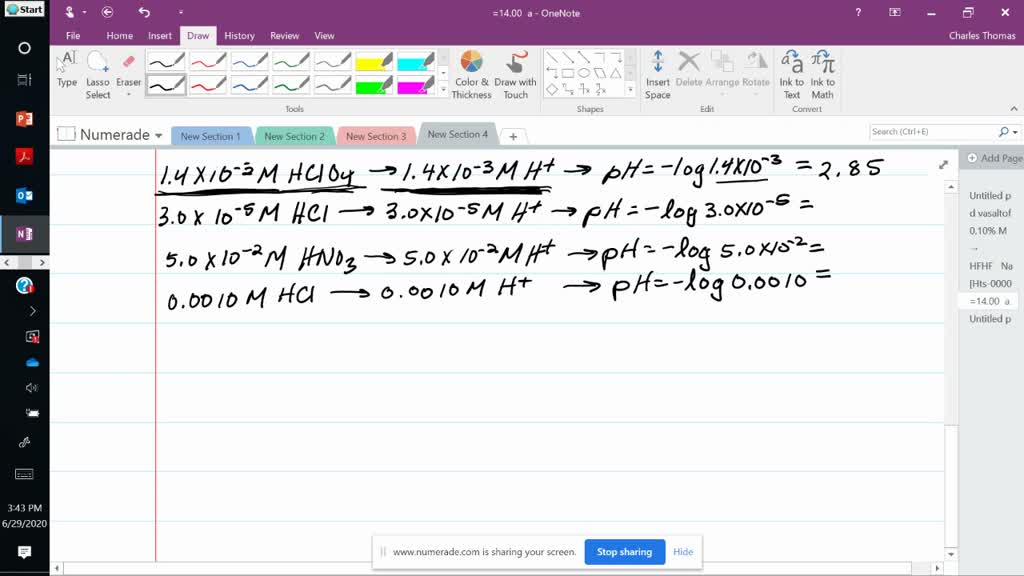
When dealing with strong acids, the pH can be calculated using a straightforward method. Strong acids completely dissociate in water, producing a high concentration of hydrogen ions (H+). This means that for strong acids like hydrochloric acid (HCl), the concentration of H+ ions directly translates to the pH value.
For example, suppose we have a 1 M solution of HCl. The pH can be calculated using the formula:
pH = -log[H+]
where [H+] is the concentration of hydrogen ions in moles per liter (M).
Substituting the values, we get:
-log(1) = 0
Therefore, the pH of a 1 M solution of HCl is 0.
Weak Acid pH Calculations
Weak acids, on the other hand, do not fully dissociate in water. This means that the concentration of hydrogen ions is less than the concentration of the weak acid. To calculate the pH of a weak acid solution, we need to use the acid dissociation constant (Ka).
The pH of a weak acid solution can be calculated using the formula:
pH = 1/2(pKa – log[A-])
where [A-] is the concentration of conjugate base ions, and pKa is the negative logarithm of the acid dissociation constant (Ka).
For example, let’s consider a 0.1 M solution of acetic acid (CH3COOH). The pKa value for acetic acid is 4.76.
First, we need to calculate the concentration of conjugate base ions ([A-]):
[CH3COO-] = 0.1 M
Next, we use the formula to calculate the pH:
pH = 1/2(4.76 – log(0.1))
Using a calculator to evaluate the expression, we get:
pH ≈ 2.88
Therefore, the pH of a 0.1 M solution of acetic acid is approximately 2.88.
Base pH Calculations
When dealing with strong bases, the pH can be calculated using a similar method to that of strong acids. However, we need to use the concentration of hydroxide ions (OH-) instead of hydrogen ions.
For example, let’s consider a 1 M solution of sodium hydroxide (NaOH). The pH can be calculated using the formula:
pH = -log[OH-]
where [OH-] is the concentration of hydroxide ions in moles per liter (M).
Substituting the values, we get:
-log(1) = 0
Therefore, the pH of a 1 M solution of NaOH is 0.
When dealing with weak bases, we need to use the base dissociation constant (Kb). The pH of a weak base solution can be calculated using the formula:
pOH = 1/2(BpKb – log[B+])
where [B+] is the concentration of conjugate acid ions, and BpKb is the negative logarithm of the base dissociation constant (Kb).
For example, let’s consider a 0.1 M solution of ammonia (NH3). The BpKb value for ammonia is 4.75.
First, we need to calculate the concentration of conjugate acid ions ([B+]):
[NH4+] = 0.1 M
Next, we use the formula to calculate the pOH:
pOH = 1/2(4.75 – log(0.1))
Using a calculator to evaluate the expression, we get:
pOH ≈ 9.92
Since pH = 14 – pOH, we can calculate the pH:
pH = 14 – 9.92
pH ≈ 4.08
Therefore, the pH of a 0.1 M solution of ammonia is approximately 4.08.
Understanding pH in Various Chemical Reactions

pH plays a crucial role in various chemical reactions, and its behavior can influence the outcome of these reactions. Understanding pH in different chemical reactions is essential for predicting and controlling the reaction outcomes.
The pH of a solution can change dramatically during chemical reactions, depending on the reactants involved. In neutralization reactions, for example, the pH can change from acidic to basic or vice versa. In precipitation reactions, the pH can affect the solubility of the reactants and products.
Neutralization Reactions
Neutralization reactions involve the combination of an acid and a base to form a salt and water. In these reactions, the pH of the solution changes as the acid and base react to form a new compound.
- In a neutralization reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH), the pH of the solution changes from acidic to basic.
- The reaction between HCl and NaOH results in the formation of sodium chloride (NaCl) and water.
- The pH of the solution changes as the acid and base react, resulting in a solution with a basic pH.
pH = -log[H+]
HCl + NaOH → NaCl + H2O
The behavior of pH in neutralization reactions is complex and can be influenced by various factors, including the concentration of the reactants and the presence of other substances in the solution.
Precipitation Reactions
Precipitation reactions involve the formation of a solid product from a solution. In these reactions, the pH of the solution can affect the solubility of the reactants and products.
- In a precipitation reaction between silver nitrate (AgNO3) and sodium chloride (NaCl), the pH of the solution affects the solubility of the reactants and products.
- The reaction between AgNO3 and NaCl results in the formation of silver chloride (AgCl) and sodium nitrate (NaNO3).
- The pH of the solution can affect the solubility of AgCl, resulting in the formation of a precipitate.
AgNO3 + NaCl → AgCl + NaNO3
The behavior of pH in precipitation reactions is also complex and can be influenced by various factors, including the concentration of the reactants and the presence of other substances in the solution.
The significance of pH in chemical reactions cannot be overstated. pH can influence the rate and outcome of reactions, making it an essential factor in understanding and predicting reaction outcomes. Understanding pH in various chemical reactions is crucial for developing new applications and improving existing ones.
Calculating pH with pH Buffer Solutions
pH buffer solutions are a crucial tool in chemistry, particularly in laboratory settings, for maintaining a stable pH environment. These solutions consist of a weak acid and its conjugate base, which work together to maintain a constant pH level. This property makes them essential in various applications, including biochemistry, pharmaceuticals, and environmental monitoring.
Properties and Applications of pH Buffer Solutions
pH buffer solutions have several distinct properties that make them useful in various fields:
- Buffer capacity: pH buffer solutions can maintain a stable pH level over a wide concentration range.
- Reversible nature: The weak acid and its conjugate base can convert reversibly to each other, enabling the solution to return to its original pH level after an external influence.
- Wide pH range: pH buffer solutions can be designed to maintain a stable pH level within a specific pH range.
These solutions find applications in various fields, including:
- Biomedical research: pH buffer solutions are used to maintain the required pH levels in biological buffer systems, ensuring the stability of biological molecules and enzymes.
- Pharmaceutical industry: pH buffer solutions are used as excipients in various medications, helping to stabilize the drug and maintain its efficacy.
- Environmental monitoring: pH buffer solutions are used to maintain a stable pH level in laboratory and field environments, allowing for more accurate analysis of environmental samples.
Calculating pH with pH Buffer Solutions
To calculate the pH of a pH buffer solution, the Henderson-Hasselbalch equation can be used:
pH = pKa + log10 ([A-]/[HA])
where:
– pKa is the acid dissociation constant of the weak acid
– [A-] is the concentration of the conjugate base (in moles per liter)
– [HA] is the concentration of the weak acid (in moles per liter)
Numerical Example
Suppose we have a pH buffer solution consisting of 0.1 M acetic acid (HA) and 0.2 M acetate ion (A-). The acid dissociation constant (pKa) of acetic acid is 4.76.
Calculate the pH of the solution.
Using the Henderson-Hasselbalch equation:
pH = 4.76 + log10 (0.2/0.1) = 4.76 + log10 (2) = 4.76 + 0.3 = 5.06
The pH of the solution is approximately 5.06.
Advantages and Limitations of Using pH Buffer Solutions
The advantages of using pH buffer solutions include:
- Stable pH levels: pH buffer solutions can maintain a stable pH level over a wide concentration range.
- Reversible nature: The weak acid and its conjugate base can convert reversibly to each other, enabling the solution to return to its original pH level after an external influence.
However, there are also limitations:
- Sensitivity to temperature and concentration: pH buffer solutions can be sensitive to changes in temperature and concentration, which can affect their pH-maintaining capacity.
- Interference from external factors: pH buffer solutions can be affected by external factors such as ionic strength, pH, and concentration of other solutes, which can interfere with their pH-maintaining capacity.
Common Mistakes in pH Calculations
Common mistakes in pH calculations can lead to inaccurate results, which may have significant consequences in various fields such as chemistry, biology, and environmental science. Incorrect assumptions and calculation errors are among the most common mistakes made when determining pH levels. In this section, we will discuss these common mistakes and provide examples of how to recognize and correct them, as well as strategies to improve the accuracy of pH calculations.
Incorrect Assumptions
Incorrect assumptions can lead to inaccurate pH calculations. One common assumption is that all chemicals are completely dissociated in water, which is not always the case. Another assumption is that the volume of the solution is known, when in fact it is not.
-
H2O does not always fully dissociate into H+ and OH− ions.
-
Inadequate Information: The amount of a particular compound may not be known exactly, leading to difficulties in calculating its concentration and hence its pH value.
Calculation Errors
Calculation errors can also occur when determining pH levels. Examples include:
-
The molarity and volume of the solution must be known and the calculation should be accurate with the correct units.
Not accounting for impurities: The presence of impurities may affect the pH levels, and their effects should be considered in the calculation.
Misuse of pH Scale
The pH scale is often misused or misunderstood, leading to incorrect conclusions. It is essential to understand that the pH scale is logarithmic, with a range of 0-14, where a pH of 7 is neutral, and values below 7 are acidic, while those above 7 are basic.
-
A pH value close to 7 may not necessarily indicate neutrality in all cases, especially in solutions with very low H+ concentrations.
-
Not considering the context: pH levels may not be relevant in certain situations, such as in strongly acidic or basic environments.
Insufficient Data
Insufficient data can lead to inaccurate pH calculations. Common issues include:
-
An unknown or uncertain pH value may impact the accuracy of further calculations, such as in the context of chemical reactions or titrations.
Lack of control samples: Inadequate data on control samples may limit the scope of the experiment, reducing the generalizability of the findings.
Equipment Malfunction
Equipment malfunction can also contribute to inaccurate pH calculations. Examples include:
-
Improper calibration of pH meters or spectrophotometers can result in erroneous readings.
-
Leaking equipment: Leaks in equipment, such as pH meters, can lead to incorrect readings due to the presence of impurities or contaminants.
pH Calculations in Environmental and Industrial Applications
pH calculations play a crucial role in various environmental and industrial applications, including water treatment and chemical processing. Accurate pH calculations are essential in these contexts, as they directly impact the efficiency, safety, and environmental sustainability of these processes.
The Importance of pH in Water Treatment
pH calculations are critical in water treatment plants, where they help determine the optimal conditions for coagulation, sedimentation, and disinfection.
pH 7.0-8.5 is the ideal range for effective coagulation and flocculation in water treatment
The pH of water affects the solubility of dissolved solids, the stability of suspended particles, and the effectiveness of disinfectants. For example, a pH range of 7.0-8.5 is considered optimal for coagulation and flocculation in water treatment, as it allows for the formation of stable flocs that can be easily removed from the water. On the other hand, high pH values can lead to the formation of insoluble precipitates that can clog pipes and equipment.
pH Calculations in Chemical Processing
pH calculations are also essential in chemical processing, where they help determine the optimal conditions for chemical reactions, including neutralization, precipitation, and oxidation.
pH 5.5-6.5 is the ideal range for optimizing chemical reactions in industrial processes
For instance, in the production of aluminum sulfate, a pH range of 5.5-6.5 is considered optimal for the reaction between sulfuric acid and aluminum hydroxide. This range allows for the formation of soluble aluminum sulfate, which can be easily separated from the reaction mixture.
Case Study: pH Management in a Power Plant
A power plant uses limestone to absorb sulfur dioxide emissions from coal combustion. The resulting gypsum is sold as a byproduct. However, if the pH of the limestone slurry is too high, it can lead to the formation of calcium carbonate, which can cause pipes to become clogged.
To manage the pH of the limestone slurry, the power plant measures the pH of the slurry at regular intervals. If the pH exceeds 11.0, they add a small amount of sulfuric acid to lower the pH. This ensures that the limestone dissolves efficiently, and the gypsum is produced in the desired form.
Case Study: pH Monitoring in a Waste Water Treatment Plant
A waste water treatment plant uses a combination of physical, chemical, and biological processes to remove pollutants from wastewater. The pH of the wastewater can affect the efficiency of these processes. For example, if the pH is too high, it can lead to the formation of nitrogenous compounds, which can cause water pollution.
To monitor the pH of the wastewater, the treatment plant uses a pH meter to measure the pH at regular intervals. If the pH exceeds 9.0, they add a small amount of sulfuric acid to lower the pH. This ensures that the wastewater is treated efficiently, and the pollutants are removed in the desired form.
Closure
In conclusion, mastering the various methods of pH calculations is essential for accurate and reliable results in various fields. By understanding the fundamental principles and applications of pH calculations, readers can make informed decisions and achieve their goals. Whether you’re a student or a professional, this guide has provided you with a comprehensive overview of pH calculations and its significance in various contexts.
FAQ Section
What is pH and why is it important?
pH is a measure of the acidity or basicity of a solution. It plays a crucial role in various biological and chemical processes, including digestion, respiration, and chemical reactions.
What is the Henderson-Hasselbalch equation?
The Henderson-Hasselbalch equation is a mathematical formula used to calculate pH from the concentrations of a weak acid and its conjugate base.
What are pH buffer solutions?
pH buffer solutions are mixtures of a weak acid and its conjugate base that resist changes in pH when small amounts of acid or base are added.
Can pH calculations be used in environmental applications?
Yes, pH calculations play a crucial role in environmental applications, including water treatment and wastewater management.
What are the common mistakes in pH calculations?
Common mistakes in pH calculations include incorrect assumptions, calculation errors, and using the wrong pH scale.