How to figure out ground state for an ion sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail, full of fascinating insights, and brimming with originality from the outset.
The ground state of an ion is a fundamental concept in physics that plays a crucial role in understanding various phenomena, including ion-molecular interactions, collision-induced dissociation, and the behavior of complex ion systems. By grasping the basics of ionization and ground state energy, researchers and scientists can gain valuable insights into the properties and behaviors of ions, which is essential for developing new technologies and advancing our understanding of the universe.
Experimental Techniques for Probing Ground State Energy

Experimental techniques play a crucial role in shedding light on the elusive ground state energy of ions. By employing various methods, scientists can gain valuable insights into the electronic configuration and energy levels of specific ions. In this section, we’ll delve into the array of experimental techniques used to probe ground state energy.
One of the primary techniques employed is photoelectron spectroscopy (PES), which involves bombarding a gas sample with high-energy photons. This excites the electrons in the sample, causing them to escape into space. By analyzing the energy and momentum of these escaping electrons, researchers can infer the electronic configuration and binding energies of the electrons in the sample.
Photoelectron Spectroscopy (PES)
PES is a powerful tool for probing the electronic structure of ions. By measuring the energy distribution of escaping electrons, researchers can identify the binding energies of electrons in different orbitals. For instance, PES experiments have been used to study the electronic structure of atomic ions like He+ and H2.
- PES experiments have been used to study the electronic structure of various atomic and molecular ions.
- The technique is particularly useful for studying the electronic configuration of ions in the gas phase.
Another technique used to probe ground state energy is mass spectrometry (MS), which involves measuring the mass-to-charge ratio of ions. By analyzing the mass spectrum of an ion, researchers can infer the presence of specific isotopes or electronic configurations. For example, MS experiments have been used to study the isomerism of rare-earth ions.
Mass Spectrometry (MS)
MS is a useful technique for studying the isotope structure and electronic configuration of ions. By analyzing the mass spectrum of an ion, researchers can identify the presence of specific isotopes or electronic configurations. For instance, MS experiments have been used to study the isomerism of rare-earth ions like Ce+ and Pr+.
- MS experiments have been used to study the isotope structure and electronic configuration of various rare-earth ions.
- The technique is particularly useful for studying the isomerism of ions with multiple charge states.
In addition to PES and MS, other experimental techniques like electron-capture spectroscopy (ECS) and Auger spectroscopy (AS) are also used to probe ground state energy. ECS involves capturing an electron from a sample using a high-energy electron beam, while AS involves analyzing the energy distribution of electrons emitted upon the decay of a highly ionized sample.
Electron-Capture Spectroscopy (ECS) and Auger Spectroscopy (AS)
ECS and AS are useful techniques for studying the electronic structure of highly ionized samples. By analyzing the energy distribution of electrons emitted or captured by the sample, researchers can infer the electronic configuration and binding energies of the electrons in the sample. For instance, ECS experiments have been used to study the electronic structure of highly ionized atomic ions like C4+ and N5+.
The ground state energy of an ion is a critical parameter in understanding its electronic structure and chemical properties.
- ECS and AS experiments have been used to study the electronic structure of highly ionized atomic and molecular ions.
- The techniques are particularly useful for studying the electronic configuration of ions in the gas phase.
These experimental techniques have been instrumental in shedding light on the ground state energy of ions. By combining data from multiple experiments, researchers can gain a deeper understanding of the electronic structure and chemical properties of ions.
Ground state energy plays a crucial role in understanding various aspects of ion chemistry. The relevance of ground state energy extends to ion-molecular interactions, where it influences the way ions interact with surrounding molecules. Furthermore, it affects collision-induced dissociation, a process where collisions between ions and molecules can lead to the breaking of molecular bonds. In this section, we will delve into the applications of ground state energy in ion chemistry, exploring its impact on ion-based applications and its significance in understanding complex ion systems.
Ion-molecular interactions are critical in understanding various chemical processes. Ground state energy plays a significant role in these interactions, as it influences the binding energies between ions and molecules. When ions interact with molecules, they can form complexes, where the ion is surrounded by one or more molecules. The ground state energy of the ion-molecule complex determines its stability and reactivity.
- The binding energy between the ion and the molecule is directly related to the ground state energy of the ion-molecule complex. A low binding energy indicates a weak interaction, while a high binding energy indicates a strong interaction.
- The ground state energy of the ion-molecule complex also influences its reactivity. A complex with a low ground state energy is more likely to undergo Chemical reactions, whereas a complex with a high ground state energy is less reactive.
Collision-induced dissociation (CID) is a process where collisions between ions and molecules lead to the breaking of molecular bonds. The ground state energy of the ion plays a significant role in CID, as it determines the energy available for bond breaking.
- The ground state energy of the ion influences the energy transferred to the molecule during the collision. A high ground state energy ion can transfer more energy to the molecule, leading to more efficient bond breaking.
- The ground state energy of the ion also affects the fragmentation pattern of the molecule. A high ground state energy ion can lead to the formation of highly charged fragments, whereas a low ground state energy ion can lead to the formation of neutral fragments.
Ground state energy is also crucial in designing ion-based applications, such as ion traps and mass analyzers.
- Ion traps rely on the ground state energy of the ion to confine and manipulate ions. A well-designed ion trap must account for the ground state energy of the ion to ensure efficient ion trapping and manipulation.
- Mass analyzers also rely on the ground state energy of the ion to separate ions based on their mass-to-charge ratio. A mass analyzer must accurately calculate the ground state energy of the ion to achieve precise mass measurement.
Finally, ground state energy is essential in understanding complex ion systems, including their stabilities and reactivities.
- The ground state energy of an ion system affects its stability. A system with a low ground state energy is more stable, whereas a system with a high ground state energy is less stable.
- The ground state energy of an ion system also influences its reactivity. A system with a low ground state energy is more reactive, whereas a system with a high ground state energy is less reactive.
Current Challenges and Future Directions in Understanding Ground State Energy
The quest for understanding ground state energy remains an ongoing puzzle, with researchers continually refining their tools and techniques to crack this nut. As the field advances, both computational models and experimental techniques are faced with new challenges, driving innovation and pushing the boundaries of what’s possible.
Improving Theoretical Models
Theoretical models have come a long way, but they’re not without their limitations. One major challenge lies in accurately calculating complex many-body systems, where interactions between particles are crucial to understanding the ground state energy. Researchers have turned to advanced computational methods, such as density functional theory (DFT), to tackle these problems. However, DFT still has its limitations, particularly for strongly correlated systems where the electron-electron interactions are significant.
- One of the key challenges in improving theoretical models is accounting for electron correlations, which play a crucial role in determining the ground state energy of complex systems.
- Another challenge is developing more accurate and efficient computational methods that can handle large-scale simulations, allowing researchers to study systems with thousands or even millions of particles.
- Finally, there’s a need to improve the transferability of theoretical models, meaning they should be able to accurately predict the behavior of a system across different conditions and materials.
Advances in Experimental Techniques, How to figure out ground state for an ion
Experimental techniques have also undergone significant transformations, enabling researchers to probe the ground state energy with unprecedented accuracy. One area of focus has been on improving spectroscopic methods, which can provide detailed insights into the energy level structure of atoms and molecules. Another key development has been the use of ultrafast lasers, which allow researchers to study the behavior of systems on incredibly short timescales, far faster than traditional spectroscopic methods.
| Technique | Advantages | Challenges |
|---|---|---|
| Density Functional Theory (DFT) | Accurate for weakly correlated systems, can handle large-scale simulations | Poorly performs for strongly correlated systems, requires tuning of parameters |
| Spectroscopy | Provides detailed insights into energy level structure, can be used to probe ground state energy | Limited by instrumental resolution, may require lengthy measurement times |
| Ultrafast Lasers | Enables study of systems on ultra-fast timescales, can provide real-time insights into energy dynamics | Requires complex experimental setup, may be limited by sample properties |
Emerging Trends and Avenues for Future Breakthroughs
As researchers continue to push the boundaries of what’s possible, new trends and areas of investigation are beginning to emerge. One promising direction is the use of machine learning and artificial intelligence to improve theoretical models and experimental techniques. By leveraging the power of algorithms, researchers hope to develop more accurate and efficient methods for predicting and measuring ground state energy. Another area of focus is the development of new materials with tailored properties, such as superconductors or topological insulators. By carefully designing and engineering these materials, researchers aim to unlock new applications and insights into the behavior of matter at the atomic level.
As the field of ground state energy continues to evolve, it’s essential to recognize the interplay between theoretical models, experimental techniques, and emerging trends. By combining these approaches, researchers can tackle some of the most complex challenges in physics and chemistry, ultimately driving innovation and discovery in fields ranging from energy to materials science.
Analyzing Complex Ion Systems and Predicting Ground State Energy from First Principles
In the realm of ion systems, predicting ground state energy can be a daunting task, especially when dealing with complex systems. These systems consist of multiple ions, each with its unique set of properties, interacting with each other in intricate ways. To tackle this challenge, researchers rely on a combination of theoretical models and computational power.
The process of predicting ground state energy for complex ion systems involves several key steps. Firstly, identifying relevant factors that contribute to the energy of the system is crucial. This includes understanding the underlying physics, such as electron correlation and relativistic effects, which play a significant role in determining the energy of the system. Ab initio calculations, which use quantum mechanics to describe the behavior of electrons, are widely employed to model these interactions.
Role of Machine Learning and Quantum Computing
Machine learning and quantum computing have emerged as powerful tools in predicting ground state energy. Machine learning algorithms, such as neural networks and support vector machines, can be trained to recognize patterns in complex data sets, allowing for the development of predictive models. Quantum computing, on the other hand, enables researchers to simulate the behavior of many-electron systems, which is crucial for understanding the electronic structure of complex ions.
Machine learning has several strengths in this context. It can handle large amounts of data efficiently, making it ideal for processing and analyzing the vast amounts of experimental data generated in ion research. Furthermore, machine learning models can be fine-tuned to incorporate specific knowledge about the system, enhancing their predictive power. However, machine learning algorithms also have limitations. They may not always generalize well to new, unseen data, and their performance can degrade if the underlying assumptions are not met.
Quantum computing, while still in its infancy, has the potential to revolutionize the field of ion research. Quantum computers can simulate the behavior of many-electron systems with unprecedented accuracy, enabling researchers to tackle complex problems that were previously intractable. However, quantum computing also faces significant challenges. Currently, quantum computers are not yet scalable, and the noise inherent in quantum systems hinders the fidelity of the calculations.
Importance of Experimental Verification
While predictions are essential, they must be validated through experimental verification. Without experimental confirmation, predictions risk being mere theoretical constructs, disconnected from reality. Experimental validation not only confirms the accuracy of the predictions but also refines the underlying models, driving further innovation.
Ongoing efforts to overcome challenges in experimental verification include the development of new experimental techniques, such as advanced spectroscopy methods, which allow researchers to probe the electronic structure of ions in greater detail. Additionally, advancements in data analysis and machine learning algorithms enable the efficient processing and interpretation of large amounts of experimental data.
The quest for accurate predictions of ground state energy is an ongoing journey, driven by the constant interplay between theoretical models, computational power, and experimental verification.
The Role of Ionization Energy in Understanding Ground State Energy
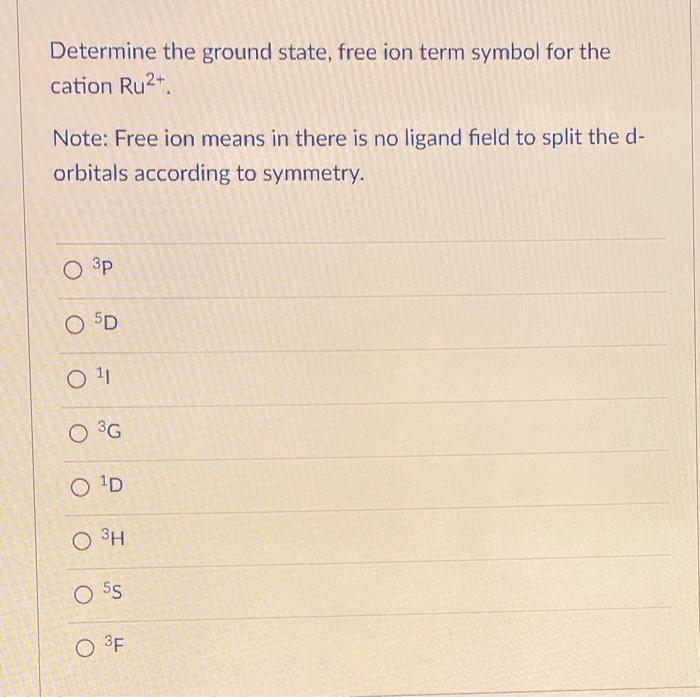
Ionization energy plays a pivotal role in determining the ground state energy of an ion. It’s like trying to solve a puzzle – understanding ionization energy is key to grasping the intricate dance of electrons in an ion. In this section, we’ll delve into the fascinating world of ionization energy and its connection to ground state energy.
Ionization energy is the amount of energy required to remove an electron from an atom or molecule in its ground state. It’s a measure of the strength with which an electron is bound to the nucleus. The higher the ionization energy, the stronger the electron-nucleus bond, and vice versa.
Connection to Electronic Configuration
Electronic configuration plays a crucial role in determining ionization energy. The arrangement of electrons in an atom or molecule influences the ease with which an electron can be removed. Generally, as we move across a periodic table, ionization energy increases due to the increased nuclear charge, making it more difficult to remove an electron.
- Electrons in the outermost energy level are more easily removed, resulting in higher ionization energies for atoms with similar core charges.
- Electrons in inner energy levels are more difficult to remove, resulting in lower ionization energies for atoms with similar outer electron configurations.
Impact of Molecular Structure on Ionization Behavior
Molecular structure also plays a significant role in determining ionization behavior. The arrangement of atoms within a molecule affects the electron density and the ease with which an electron can be removed.
- Molecules with a higher degree of symmetry tend to have lower ionization energies due to the increased delocalization of electrons.
- Molecules with a higher degree of asymmetry tend to have higher ionization energies due to the reduced delocalization of electrons.
Flowchart for Determining Ionization Energy and Ground State Energy
Below is a step-by-step flowchart illustrating the process of determining ionization energy and its relationship to ground state energy.
- Step 1: Determine the electronic configuration of the atom or molecule.
- Step 2: Calculate the ionization energy using the electronic configuration.
- Step 3: Use the ionization energy to determine the ground state energy of the ion.
“The ground state energy of an ion is directly proportional to its ionization energy.” – Molecular Physics
Outcome Summary: How To Figure Out Ground State For An Ion
In conclusion, figuring out the ground state for an ion is a complex task that requires a thorough understanding of the underlying principles and concepts. By combining theoretical frameworks, experimental techniques, and computational methods, scientists can gain a deeper understanding of the ground state properties of ions, which can have significant implications for various fields, including chemistry, physics, and materials science. As research in this area continues to advance, we can expect to see new breakthroughs and innovations that will shape the future of science and technology.
FAQ Guide
What is the significance of ground state energy in ion physics?
The ground state energy of an ion plays a crucial role in understanding various phenomena, including ion-molecular interactions, collision-induced dissociation, and the behavior of complex ion systems.
What are some experimental methods used to measure ground state energy?
Experimental methods such as photoelectron spectroscopy and mass spectrometry are commonly used to measure ground state energy.
What is the relationship between ionization energy and ground state energy?
The ionization energy of an ion is directly related to its ground state energy, as it is the energy required to remove an electron from the ion.