With how to find density at the forefront, this article delves into the fascinating world of physics, where measurements and calculations play a crucial role in understanding the behavior of matter. The concept of density is not just a random variable, but rather a vital tool that helps us comprehend the properties and characteristics of various substances. As we embark on this journey to explore how to find density, we’ll uncover the intricacies of density measurements, the factors that affect them, and most importantly, the practical applications of this fundamental concept.
We’ll discuss the differences betweenbulk density, true density, and apparent density, their real-world applications, and the various scientific instruments used to measure density. Additionally, we’ll delve into the factors that affect density calculations, such as temperature, pressure, and atmospheric conditions, as well as the impact of molecular structure and atomic composition on the resulting density values. By the end of this article, you’ll possess a comprehensive understanding of how to find density with accuracy and how to apply this knowledge in real-world scenarios.
Utilizing Scientific Instruments for Density Measurements
Density measurements are crucial in various fields, including chemistry, physics, and engineering. Scientific instruments play a vital role in determining the density of materials, and in this section, we will explore the operation and function of hydrometers, pycnometers, and balance scales.
Hydrometers for Density Measurements
Hydrometers are calibrated containers that measure the density of fluids. They work on the principle that the buoyancy force exerted on an object by a fluid is proportional to the density of the fluid. A hydrometer consists of a weighted glass bulb, a stem, and a graduated scale. Here’s how it works:
-
Fill the hydrometer’s container with the fluid to be measured, making sure the graduations are visible.
- The buoyancy force exerted on the hydrometer causes it to rise to a certain level in the fluid.
- Determine the temperature of the fluid by placing the hydrometer in a calibrated thermometer.
- The reading on the graduated scale corresponds to the density of the fluid at the given temperature.
Hydrometers are suitable for measuring the density of clear, colorless liquids with a low viscosity. However, they are not suitable for thick or viscous fluids.
Pycnometers for Density Measurements
Pycnometers are another type of instrument used for density measurements. They consist of a cylindrical container and a cap with a narrow neck. Here’s how they work:
-
Filling the pycnometer container with the material (solid or liquid) to be measured.
- The volume of the material is then measured by the known volume of the pycnometer.
- Next, the mass of the pycnometer and its contents is weighed on a balance scale.
- The density of the material can be calculated using the formula: density = (mass / volume)
Pycnometers are more accurate than hydrometers and can be used for a wider range of materials, including powders and granules.
Balance Scales for Density Measurements
Balance scales measure the mass of a substance by balancing it with known weights. The density of a substance can be calculated using the formula: density = mass / volume. Here’s how to use a balance scale:
-
Place the pycnometer containing the material on the balance scale.
- Determine the mass of the pycnometer and its contents by reading the balance scale.
- Measure the volume of the material using a graduated container.
- The density of the material can be calculated using the formula: density = (mass / volume)
Balance scales are more accurate than hydrometers and pycnometers, especially for low-density materials.
Advantages and Limitations of Each Instrument
Each instrument has its own advantages and limitations:
-
Hydrometers:
- Advantages:
-
Easy to use and inexpensive.
- Accurate for measuring the density of clear, colorless liquids.
- Limited to thin liquids and not suitable for thick or viscous fluids.
-
Pycnometers:
- Advantages:
-
Accurate for a wide range of materials, including powders and granules.
- Avoids the need for a balance scale or other instruments.
- Durable and resistant to corrosion.
- Limited to measuring the density of a single sample.
-
Balance Scales:
- Advantages:
-
Highly accurate for measuring the density of low-density materials.
- Can be used for a wide range of materials, including powders and granules.
- No need for calibration or adjustment.
- Expensive and requires a significant amount of expertise to use.
In conclusion, each instrument has its own advantages and limitations, and the choice of instrument depends on the specific application and material being measured.
Performing Density Calculations in the Laboratory: How To Find Density
Density calculations are a crucial step in the laboratory, as they help scientists determine the mass and volume of various substances. Accurate density measurements are essential in fields such as materials science, chemistry, and physics, where the properties of materials are being studied. By performing density calculations, researchers can gain a deeper understanding of the characteristics of a substance and its behavior under different conditions.
In the laboratory, density calculations can be performed using various formulas, each with its own specific variables and applications. The true density of a substance, for example, is often calculated using the formula:
True Density = Mass / True Volume
Another common formula used to calculate density is the mass-per-unit-volume (mpv) method, which is expressed as:
mpv = Mass / Volume
The Archimedes’ Principle, on the other hand, is used to calculate the density of a substance by measuring its buoyancy in water. This method is expressed as:
Density = Mass / Displacement
### Calculating True Density
True density is a critical measure of a substance’s density, as it takes into account the presence of any voids or pores within the material. To calculate true density, researchers must use a procedure known as helium pyknometry or gas displacement pyknometry.
- Measure the mass of the substance using a balance or scale.
- Measure the volume of the substance using a gas displacement pipette or a volume meter.
- Calculate the true density by dividing the mass by the true volume.
### Comparing Density Calculations
The following table compares the results of different density calculations:
| Formula | Resulting Formula | ||
|---|---|---|---|
| True Density = Mass / True Volume | Mass = 100 g | True Volume = 10 cm^3 | 3.00 g/cm^3 |
| mpv = Mass / Volume | Mass = 50 g | Volume = 100 cm^3 | 0.50 g/cm^3 |
| Density = Mass / Displacement | Mass = 200 g | Displacement = 50 cm^3 | 4.00 g/cm^3 |
In this table, the true density of a substance is calculated as 3.00 g/cm^3, while the mpv method yields a result of 0.50 g/cm^3. The Archimedes’ Principle also gives a density of 4.00 g/cm^3.
By understanding the different methods of density calculations, researchers can select the most suitable approach for their specific research needs.
Interpreting and Applying Density Data in Scientific Research
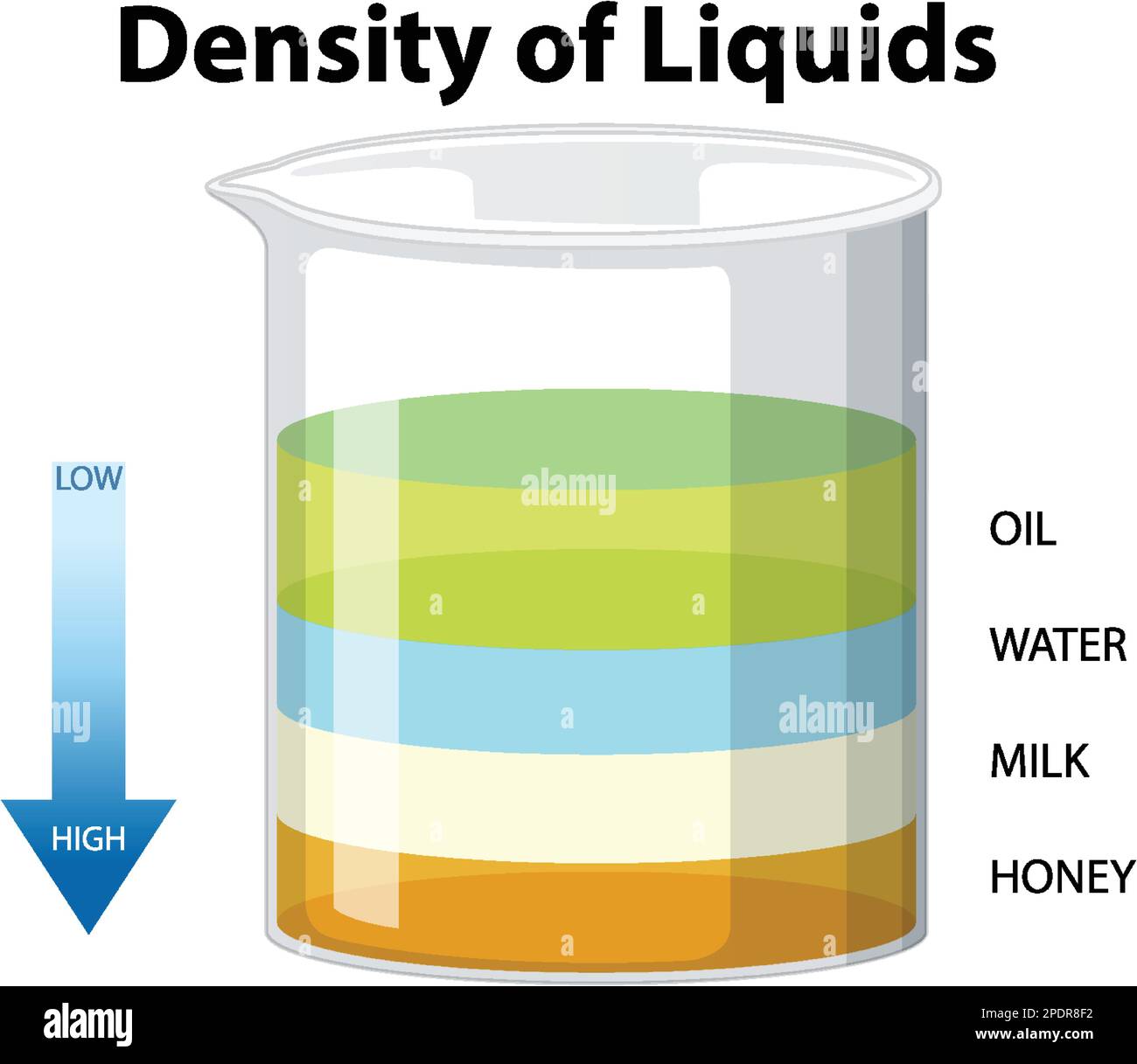
Density data plays a vital role in various scientific fields, serving as a fundamental characteristic that influences the properties and behavior of materials in different environments. By understanding the density of a substance, researchers can accurately predict its behavior under diverse conditions, such as changes in temperature and pressure. This knowledge is essential for designing and optimizing materials for various applications, from engineering materials to pharmaceuticals.
Understanding Material Properties
Density is a critical parameter in determining the material properties of substances, including their stability, reactivity, and thermal conductivity. For instance, a higher density material tends to have a higher melting and boiling point, whereas a lower density material is more likely to have a lower melting and boiling point. This knowledge is invaluable in various industries, including metallurgy, ceramics, and polymer science.
Some key material properties influenced by density include:
- Thermal conductivity: Density affects the ability of a material to conduct heat. Materials with higher density tend to have higher thermal conductivity.
- Compressibility: Density influences a material’s compressibility, with higher density materials being less compressible.
- Reactivity: Density can impact a material’s reactivity, with higher density materials being more resistant to chemical reactions.
These material properties have significant implications in various scientific disciplines, such as:
- Catalysis: Understanding the density of catalyst materials is crucial for optimizing their performance and efficiency.
- Materials selection: Accurate density data helps engineers and scientists select the most suitable materials for specific applications, ensuring optimal performance and safety.
- Pharmaceuticals: Density is a critical parameter in the development of pharmaceuticals, as it affects the solubility and stability of active ingredients.
Understanding the Behavior of Objects in Different Environments
Density is also essential in understanding the behavior of objects in various environments, including those subject to changes in temperature, pressure, and gravity. For example, a high-density material is less likely to behave nonlinearly under different environmental conditions, whereas a low-density material may exhibit more pronounced nonlinear behavior.
Some key examples of objects exhibiting nonlinear behavior due to density include:
- Fluid dynamics: Density affects the flow behavior of fluids, with higher density fluids being more resistant to turbulence.
- Geophysics: Understanding the density of the Earth’s crust and mantle is crucial for accurately modeling seismic activity and plate tectonics.
- Aerospace engineering: Density plays a vital role in the optimization of aircraft and satellite design, as it affects their aerodynamics and stability.
Hypothetical Experiment: Demonstration of Accuracy in Density Measurements, How to find density
To demonstrate the importance of accurate density measurements, we can design a hypothetical experiment to compare the physical properties of two materials with similar but not identical densities.
Materials and Equipment
* Material 1: A high-density metal alloy (specific gravity = 8.0)
* Material 2: A low-density polymer (specific gravity = 1.5)
* Thermometer
* Hydrometer
* Balances
* Stopwatch
Procedure
1. Measure the initial temperature of both materials using a thermometer.
2. Weigh the materials using a balance and record their weights.
3. Measure the volume of each material using a hydrometer.
4. Calculate the density of each material using the formula: density = mass/volume.
5. Repeat steps 1-4 for different environmental conditions, such as varying temperatures and pressures.
6. Compare the physical properties of both materials, such as thermal conductivity, compressibility, and reactivity.
Expected Results
* The high-density metal alloy (Material 1) will exhibit higher thermal conductivity, compressibility, and reactivity compared to the low-density polymer (Material 2).
* Material 1 will also demonstrate less nonlinear behavior under different environmental conditions, whereas Material 2 may exhibit more pronounced nonlinear behavior.
* The accurate density measurements will enable researchers to accurately predict and understand the behavior of both materials under various conditions.
This hypothetical experiment highlights the significance of accurate density measurements in understanding material properties and behavior in different environments. By acknowledging the importance of density data, researchers can make informed decisions when designing and optimizing materials for various applications, from engineering materials to pharmaceuticals.
Last Point

Now that we’ve explored the intricacies of density measurements and calculations, it’s time to take a step back and appreciate the significance of this fundamental concept. Density is an essential tool that helps us understand the properties and characteristics of various substances, allowing us to design and develop innovative products that meet specific needs. Whether you’re a student, scientist, or simply someone who’s curious about the world around us, understanding how to find density is a crucial skill that opens doors to new discoveries and applications. As we conclude this article, we hope you’ve gained a deeper appreciation for the concept of density and its importance in our daily lives.
Remember, the art of finding density is not just about throwing numbers and formulas around; it’s about understanding the underlying principles and applying them in practical scenarios. We encourage you to continue exploring the world of physics and density, and we’re confident that you’ll find it as fascinating as we do.
FAQ Insights
Q: What is the difference between bulk density and true density?
A: Bulk density is the mass of a substance per unit volume, while true density is the mass of a substance per unit volume at standard conditions.
Q: How do you calculate apparent density?
A: Apparent density is calculated by dividing the mass of a substance by its volume, but it’s affected by the presence of air pockets or impurities.
Q: What are the limitations of using a hydrometer to measure density?
A: Hydrometers are prone to errors due to temperature fluctuations, and they’re not suitable for measuring dense or viscous substances.