How to find molar mass sets the stage for this engaging narrative, offering readers a glimpse into a world where the tiny details make a big difference. Molar mass, a vital concept in chemistry, is the sum of the atomic masses of all the atoms in a molecule. But why is it so crucial? Let’s dive in and explore the fascinating world of molar mass.
In this comprehensive guide, we’ll take you through the step-by-step process of calculating molar mass, explaining the importance of atomic mass, and sharing real-world examples of how molar mass impacts chemical behavior. Whether you’re a student, teacher, or simply someone curious about chemistry, this article is tailored to meet your needs.
Calculating Molar Mass Using Atomic Mass and Molecular Formula

Calculating molar mass is a fundamental concept in chemistry that involves understanding the relationship between atomic mass, molecular mass, and the structure of compounds. The molar mass of a compound is the mass of one mole of that compound, and it can be calculated using the atomic masses of its constituent elements and the molecular formula of the compound.
In this section, we will explore how to calculate molar mass using atomic mass and molecular formula, and discuss the differences between atomic mass and molecular mass. We will also explain why atomic mass is crucial in accurately determining molar mass.
Atomic Mass and Molecular Mass
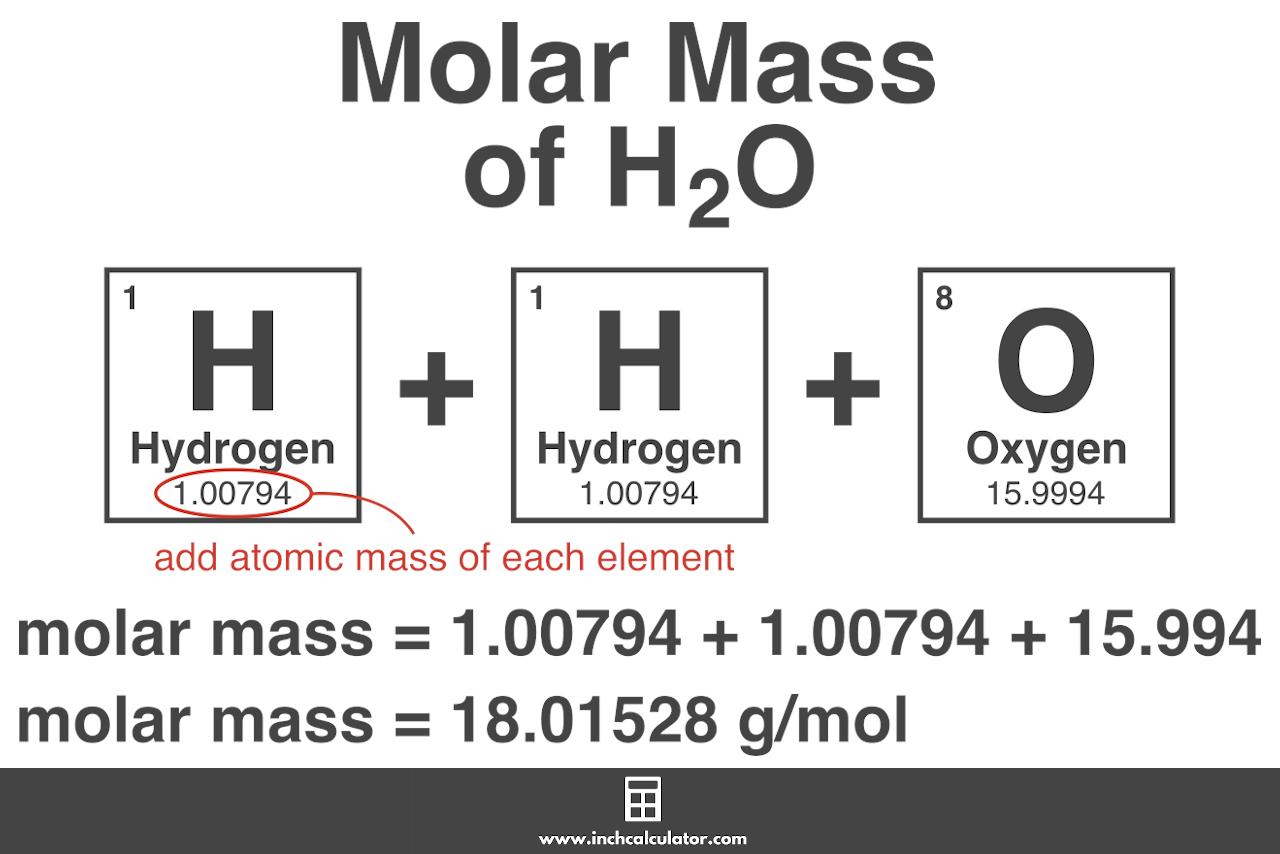
The atomic mass of an element is the average mass of a single atom of that element, including all its naturally occurring isotopes. However, the molecular mass of a compound is the sum of the atomic masses of its constituent atoms, taking into account the number of atoms of each element present. For example, the molecular mass of carbon dioxide (CO2) is 44 g/mol, which is the sum of the atomic mass of carbon (12 g/mol) and two times the atomic mass of oxygen (2 x 16 g/mol).
Calculating Molar Mass
There are several steps involved in calculating molar mass using atomic mass and molecular formula. Here’s a step-by-step guide:
### Sub-step: Determine the Molecular Formula
The molecular formula of a compound is a representation of its structure, indicating the number of atoms of each element present. For example, the molecular formula of glucose is C6H12O6, indicating that it is composed of 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms.
### Sub-step: Determine the Atomic Masses
The atomic masses of elements are listed in the periodic table. For example, the atomic mass of carbon is 12 g/mol, while that of hydrogen is 1 g/mol.
### Sub-step: Calculate the Molecular Mass
Using the molecular formula and the atomic masses of the elements, we can calculate the molecular mass of the compound. For example, if we take glucose as our example, we would calculate the molecular mass as follows:
Carbon: 6 x 12 g/mol = 72 g/mol
Hydrogen: 12 x 1 g/mol = 12 g/mol
Oxygen: 6 x 16 g/mol = 96 g/mol
Total molecular mass: 72 g/mol + 12 g/mol + 96 g/mol = 180 g/mol
Why Atomic Mass is Crucial
Atomic mass is crucial in accurately determining molar mass because it takes into account the natural isotopic composition of the elements. If an element has multiple isotopes, its atomic mass will reflect the average mass of those isotopes. If we were to calculate molar mass using the average atomic mass of an element (without considering its isotopic composition), we would arrive at an incorrect result.
For example, consider the element chlorine. Its atomic mass is 35.45 g/mol, which reflects the average mass of its two naturally occurring isotopes: 35Cl and 37Cl. If we were to calculate the molar mass of sodium chloride (NaCl) using the average atomic mass of chlorine (instead of its actual atomic mass), we would arrive at an incorrect result.
Importance of Molar Mass
Calculating molar mass is important for several reasons:
– It allows us to determine the mass of one mole of a compound, which is crucial in stoichiometry and chemical reactions.
– It helps us understand the relationship between the number of atoms of each element present in a compound and its mass.
– It enables us to compare the relative masses of different compounds.
Determining Molar Mass of Biological Molecules
Determining the molar mass of biological molecules can be a complex and challenging task due to their large size and diverse composition. Unlike simple molecules, biological molecules are often made up of many smaller units, such as amino acids, sugars, and nucleotides, which are linked together in a specific sequence. This makes it difficult to predict or measure their molar mass accurately.
Challenges in Determining Molar Mass, How to find molar mass
The unique composition and structure of biological molecules pose several challenges when it comes to determining their molar mass. For example, many biological molecules are composed of many identical subunits, which can make it difficult to accurately measure their molar mass using traditional methods such as gel electrophoresis or mass spectrometry.
Techniques Used to Measure Molar Mass in Biological Systems
Gel electrophoresis and mass spectrometry are two common techniques used to measure the molar mass of biological molecules. Gel electrophoresis separates molecules based on their size and charge, allowing researchers to estimate their molar mass based on their migration through a gel. Mass spectrometry, on the other hand, uses a beam of ions to separate and identify molecules based on their mass-to-charge ratio. Both techniques provide a reliable way to measure the molar mass of biological molecules.
Common Biological Molecules and their Molar Masses
Many biological molecules play crucial roles in various cellular processes, and understanding their molar mass is essential to understanding their function. Here are a few examples of common biological molecules, along with their calculated molar masses:
- Glucose: 180.16 g/mol
Glucose is a simple sugar that serves as a primary energy source for cells.
- Deoxyribonucleic acid (DNA): The molar mass of DNA varies depending on its sequence and length. For example, the molar mass of the human genome is approximately 3.2 x 10^9 g/mol.
DNA is a crucial molecule that carries genetic information from one generation to the next.
- Amino acid: The molar mass of an amino acid depends on its molecular structure. For example, the molar mass of glycine (the simplest amino acid) is 75.07 g/mol.
Amino acids are the building blocks of proteins, which perform a wide range of functions in cells.
- Ribonucleic acid (RNA): The molar mass of RNA depends on its sequence and length. For example, the molar mass of a typical protein-coding mRNA molecule is approximately 10^5 to 10^6 g/mol.
RNA plays a crucial role in protein synthesis and other cellular processes.
Outcome Summary
And there you have it, folks – a comprehensive guide on how to find molar mass. Whether you’re calculating molar mass from atomic mass and molecular formula or identifying molecular formulas from molar mass, we’ve got you covered. Thanks for joining me on this journey into the world of molar mass!
So, next time you’re faced with a complex chemical reaction or trying to understand the intricacies of molecular structures, remember: molar mass is the key to unlocking it all. Happy calculating!
FAQ Insights: How To Find Molar Mass
Q: What is molar mass and why is it important?
Molar mass is the sum of the atomic masses of all the atoms in a molecule. It’s crucial in defining the properties of a substance and plays a vital role in understanding chemical behavior.
Q: How do I calculate molar mass from atomic mass and molecular formula?
To calculate molar mass, multiply the atomic mass of each element in the molecular formula by its corresponding number of atoms, and then add them all together.
Q: What are some real-world scenarios where molar mass is crucial?
Molar mass impacts chemical behavior in various ways, such as affecting a substance’s density, melting point, boiling point, and solubility.
Q: Can online resources help me determine molar mass?
Yes, online tools and software can calculate molar mass for you. However, manual calculations are still essential for understanding the underlying principles.