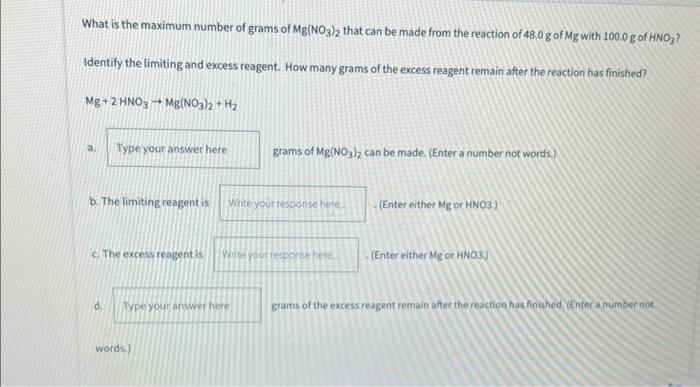
With how to find reagent in excess at the forefront, laboratories around the world are taking a much closer look at their protocols and regulations, acknowledging a reality where reagents in excess pose significant risks to the environment, people, and laboratory operations. This shift towards a more cautious approach is fueled by an increasing awareness of the potential consequences of mishandling excess reagents.
Excess reagents can stem from various factors, including spills, improper storage, and miscalculations during chemical reactions. The improper handling of these substances can lead to accidents, damage equipment, contaminate the environment, and create financial burdens. Laboratories are recognizing the need to invest in more effective tools, processes, and training for staff to mitigate these risks.
Identifying Methods for Finding Excess Reagents in a Laboratory
When working with reagents in a laboratory, it is crucial to identify and quantify the amount of reagent used to ensure the accuracy and reliability of experimental results. Excess reagents can lead to contamination, waste, and even safety hazards if not properly disposed of.
Steps for Conducting a Thorough Search for Excess Reagents
A thorough search for excess reagents involves a combination of observational techniques, measurement methods, and analytical tools. Here are the steps necessary for conducting a comprehensive search:
- Visual Inspection: Start by conducting a visual inspection of the laboratory area, equipment, and containers to identify any obvious signs of excess reagents.
- Check Labels and Records: Review labels on containers, equipment, and records to identify the quantity and type of reagents used.
- Measurement and Weighing: Use measuring instruments and balances to quantify the amount of reagents used.
- Sampling and Analysis: Collect representative samples of reagents and perform analytical tests to determine their concentration and quantity.
- Dispose of Excess Reagents Properly: Properly dispose of excess reagents according to laboratory protocols and regulations.
Commonly Used Methods for Detecting Excess Reagents
Several methods are commonly used to detect excess reagents, each with its own limitations and effectiveness.
Spectrophotometry
Spectrophotometry involves measuring the absorbance or transmittance of light through a sample to determine the concentration of reagents. This method is useful for detecting excess reagents in solutions and suspensions.
- Advantages: Highly sensitive, accurate, and can be used for a wide range of reagents.
- Limitations: Requires specialized equipment, can be time-consuming, and may not be suitable for all types of reagents.
Chromatography
Chromatography involves separating and analyzing the components of a mixture to identify and quantify excess reagents. This method is useful for detecting excess reagents in complex mixtures.
- Advantages: Highly sensitive, accurate, and can be used for a wide range of reagents.
- Limitations: Requires specialized equipment, can be time-consuming, and may not be suitable for all types of reagents.
Titration
Titration involves adding a known amount of reagent to a sample until a desired endpoint is reached to determine the concentration of excess reagents. This method is useful for detecting excess reagents in solutions.
- Advantages: Highly accurate, can be used for a wide range of reagents, and requires minimal equipment.
- Limitations: Can be time-consuming, may not be suitable for all types of reagents, and requires accurate calibration.
Semiconductor-based Sensors
Semiconductor-based sensors involve using materials with unique properties to detect excess reagents. This method is useful for detecting excess reagents in solutions.
- Advantages: Highly sensitive, accurate, and can be used for a wide range of reagents.
- Limitations: Requires specialized equipment, can be expensive, and may not be suitable for all types of reagents.
Utilizing Chemical Tests to Identify Excess Reagents
Chemical tests play a vital role in identifying excess reagents in a laboratory setting. These tests involve the use of specific reagents or indicators that react with the excess reagent in a predictable and measurable way. By utilizing chemical tests, scientists can accurately determine the presence of excess reagents and take corrective action to prevent contamination or other issues.
Using Acid-Base Titration to Detect Excess Reagents
Acid-base titration is a widely used chemical test for identifying excess reagents. This method involves the use of a strong acid or base that is slowly added to the sample until it reaches the endpoint, where the acid and base are in equilibrium. The excess reagent is then identified by the type of acid or base used. For example, using sodium hydroxide (NaOH) to titrate an acidic sample can help identify the presence of excess hydrochloric acid (HCl).
NaOH + HCl → NaCl + H2O
To perform acid-base titration, the following steps are followed:
- Prepare a stock solution of the standard acid or base.
- Prepare a sample solution containing the excess reagent.
- Slowly add the standard acid or base to the sample solution until it reaches the endpoint, as indicated by a color change of the indicator or a pH meter reading.
- Record the volume of the standard acid or base used to reach the endpoint.
Chemical Tests for Specific Reagents
Two common chemical tests used to identify specific reagents are:
Test 1: Benedict’s Test for Reducing Sugars
This test is used to identify reducing sugars, such as glucose or fructose. The test involves mixing the sample with Benedict’s reagent, which contains copper(II) sulfate. The mixture is then heated, causing the copper(II) ions to reduce to copper(I) ions, which forms a blue precipitate. The presence of a blue precipitate indicates the presence of reducing sugars.
CuSO4 + C6H12O6 → Cu2(C6H10O5)2
To perform Benedict’s test, the following steps are followed:
- Prepare a sample solution containing the suspected reducing sugar.
- Mix 1-2 drops of Benedict’s reagent with the sample solution.
- Heat the mixture gently in a water bath for 5-10 minutes.
- Observe the mixture for a blue precipitate.
Test 2: Tollens’ Test for Aldehydes
This test is used to identify aldehydes, such as formaldehyde. The test involves mixing the sample with Tollens’ reagent, which contains silver nitrate. The mixture is then reduced, causing the silver ions to form a silver mirror. The presence of a silver mirror indicates the presence of aldehydes.
CH3CHO + AgNO3 → AgCH3COO + HNO3
To perform Tollens’ test, the following steps are followed:
- Prepare a sample solution containing the suspected aldehyde.
- Mix 1-2 drops of Tollens’ reagent with the sample solution.
- Heat the mixture gently in a water bath for 5-10 minutes.
- Observe the mixture for a silver mirror.
Calibrating and Validating Chemical Tests
Before using chemical tests to identify excess reagents, it is essential to calibrate and validate them to ensure accuracy. Calibration involves adjusting the test conditions to ensure that the results are reproducible and reliable. Validation involves checking the test results against known standards or reference materials.
Calibration: Adjusting the test conditions to ensure accurate results (e.g., adjusting the pH or concentration of the reagents).
Validation: Checking the test results against known standards or reference materials (e.g., comparing the test results with the expected results from a known standard).
To calibrate and validate chemical tests, the following steps are followed:
- Determine the test conditions required for the specific chemical test (e.g., pH, temperature, or concentration of the reagents).
- Prepare a series of calibration samples with known concentrations of the excess reagent.
- Perform the chemical test on each calibration sample and record the results.
- Plot a calibration curve using the recorded results and known concentrations of the excess reagent.
- Validate the chemical test by comparing the test results with known standards or reference materials.
Safety Precautions When Handling Excess Reagents
Handling excess reagents in a laboratory can be hazardous if proper safety protocols are not followed. Laboratory personnel must be aware of the potential risks associated with these chemicals and take necessary precautions to minimize harm. This includes wearing personal protective equipment (PPE), following standard operating procedures, and being prepared for potential emergencies.
Design of Safety Protocols
A set of safety protocols should be established and communicated to laboratory personnel before handling excess reagents. These protocols should include:
- Preparation of the laboratory space: Ensure that the laboratory is well-ventilated and cleaned of any debris or hazardous materials before handling excess reagents.
- Preparation of laboratory personnel: Ensure that laboratory personnel have received proper training on the handling and disposal of excess reagents.
- Labeling of excess reagents: Label all excess reagents clearly and accurately, including their chemical names, concentrations, and hazards.
- Segregation of excess reagents: Separate excess reagents from other chemicals and materials to prevent accidents and contamination.
- Disposal of excess reagents: Establish a clear protocol for disposing of excess reagents, including the use of specialized containers and disposal procedures.
Proper communication and documentation of these protocols are crucial to ensuring the safety of laboratory personnel. Laboratory personnel handling excess reagents should always wear personal protective equipment (PPE), including: PPE should be chosen and used based on the specific hazards associated with the excess reagents being handled. Laboratory personnel should be prepared to respond to spills or leaks of excess reagents by following these emergency procedures: Regular training and practice of these emergency procedures are essential to ensuring a safe and prompt response to spills or leaks of excess reagents. Proper management and disposal of excess reagents are crucial to maintaining a safe and efficient laboratory environment. Excess reagents can pose hazards to laboratory personnel, contaminate equipment and surrounding areas, and potentially harm the environment. To address this issue, a comprehensive plan must be put in place for collecting, storing, segregating, labeling, and disposing of excess reagents. A well-structured plan is essential for managing excess reagents. This plan should include regular checks on laboratory inventory, labeling of excess reagents, and storage in designated areas. The plan should also take into account the type and quantity of reagents being used, as well as the specific disposal requirements for each reagent. Consider implementing a system for tracking and recording excess reagent storage, including container size, quantity, and location. Segregation and labeling of excess reagents are critical steps in ensuring safe disposal. Excess reagents should be grouped by category, such as chemicals, biological materials, and radioactive substances, and labeled accordingly. Labeling should include clear instructions on handling, storage, and disposal, as well as any necessary warnings or cautions. Consider implementing a standardized labeling system to ensure consistent and clear communication. Minimizing the generation of excess reagents is essential for reducing waste and minimizing the need for disposal. Consider the following strategies:
By implementing a well-structured plan for collecting, storing, segregating, and labeling excess reagents, and adopting strategies for minimizing their generation, labs can reduce waste, lower costs, and enhance safety.
In today’s laboratory settings, managing excess reagents has become increasingly important due to issues of safety, waste management, and cost savings. With digital tools, laboratories can efficiently track and manage excess reagents. This has led to the development of a range of digital tools that aid in the organization and management of reagents, reducing the workload of laboratory staff and minimizing errors. Digital Laboratory Information Management Systems (LIMS) have become a vital tool for tracking and managing excess reagents in laboratory settings. These systems help to track inventory, monitor expiration dates, and detect discrepancies in inventory levels. A LIMS can also provide critical information for regulatory compliance and ensure that laboratories maintain accurate records of chemical inventory and usage. Some of the key features of LIMS relevant to tracking excess reagents include: According to a study published in the Journal of Laboratory Automation, LIMS implementation can lead to a 30% reduction in waste and a 25% decrease in inventory costs within the first year of use. Several online platforms have been developed to aid in the management and tracking of chemical inventory and excess reagents. Some of the key platforms include: The implementation of digital tools in laboratory settings has numerous benefits, including: However, the implementation of digital tools also comes with some potential drawbacks, including: In the laboratory setting, managing excess reagents is not only a safety concern but also subject to various regulatory requirements and guidelines. Adhering to these regulations is crucial to prevent accidents, ensure a healthy work environment, and maintain compliance with laws and regulations. The handling and disposal of excess reagents are subject to stringent regulations, primarily aimed at ensuring public safety, environmental protection, and compliance with laws. These regulations vary across different jurisdictions, but a few key provisions are universally recognized as essential. Some of the key regulatory requirements and guidelines include the following: Maintaining accurate records and documentation for excess reagents is critical to ensuring regulatory compliance. Proper documentation enables laboratory personnel to track the handling and disposal of excess reagents, facilitating a seamless audit process. It also helps in identifying potential risks and hazards associated with excess reagents, thereby promoting a safe working environment. Several regulatory agencies worldwide enforce standards for managing excess reagents, ensuring public safety, environmental protection, and compliance with laws. Some examples of such agencies include: Implementing an effective laboratory inventory system is crucial for maintaining accurate records of reagents, reducing waste, and ensuring regulatory compliance. A well-designed inventory system enables laboratory managers to track and monitor chemical inventory, detect potential issues, and make informed decisions. To establish a robust laboratory inventory system, it’s essential to follow these best practices: The design of an inventory system should be tailored to the specific needs of the laboratory. Key considerations include: A well-designed system should also include features such as automated reminders, customizable reports, and easy data import/export capabilities. To ensure accurate and up-to-date records, laboratories should: To maintain an accurate inventory, laboratories should: By following these best practices, laboratories can establish a robust inventory system that ensures accurate records, reduces waste, and promotes regulatory compliance. The disposal of excess reagents poses a significant threat to the environment due to the potential release of hazardous chemicals into the ecosystem. In addition to the environmental impact, laboratories also face regulatory pressures to reduce waste generation and adopt sustainable practices. As a result, there is a growing need for laboratories to implement environmentally friendly practices for managing excess reagents. A sustainable waste management program for excess reagents involves several key components, including waste reduction, reuse, and recycling. Laboratories should first aim to reduce waste generation by minimizing reagent usage and optimizing chemical reactions. Additionally, they can explore opportunities for reusing excess reagents in other experiments or donating them to other researchers. The implementation of a sustainable waste management program requires a collaborative effort from laboratory staff, administrators, and regulatory agencies. Key stakeholders should work together to develop policies and procedures for managing excess reagents, including guidelines for waste collection, storage, and disposal. Several laboratories have successfully implemented environmentally friendly practices for managing excess reagents. For instance, the University of California, Berkeley’s College of Chemistry has a comprehensive waste management program that includes recycling, composting, and donation of excess reagents to other researchers. Similarly, the Lawrence Berkeley National Laboratory (LBNL) has implemented a Zero-Waste policy, which aims to eliminate waste generation from laboratory operations. The policy includes a range of strategies, including reducing reagent usage, reusing chemicals, and recycling or composting waste. The implementation of sustainable waste management practices for excess reagents offers several benefits, including reduced environmental impact, cost savings, and improved regulatory compliance. By minimizing waste generation and adopting environmentally friendly practices, laboratories can contribute to a more sustainable and environmentally responsible research environment. In conclusion, managing excess reagents effectively is not only essential for maintaining laboratory safety but also crucial in minimizing risks, promoting sustainability, and adhering to regulatory guidelines. What are common hazards associated with excess reagents? Spills, skin contact, and inhalation of toxic vapors are major hazards when dealing with excess reagents. How to detect excess reagents in a laboratory? Regular inventories, labeling, and chemical tests can help detect and identify excess reagents. Why is proper labeling of excess reagents crucial? Accurate labeling helps prevent mix-ups, ensures correct storage and disposal procedures, and promotes efficient inventory management.
Strategies for Managing and Disposing of Excess Reagents
Organizing a Plan for Collecting and Storing Excess Reagents
Segregating and Labeling Excess Reagents for Proper Disposal
Strategies for Minimizing the Generation of Excess Reagents
Using Digital Tools to Track and Manage Excess Reagents
Digital Laboratory Information Management Systems (LIMS) for Tracking Excess Reagents
Online Platforms for Managing and Tracking Chemical Inventory and Excess Reagents
Benefits and Drawbacks of Implementing Digital Tools in Laboratory Settings
Regulatory Compliance and Excess Reagents
Regulatory Requirements and Guidelines Governing Excess Reagents
Importance of Maintaining Accurate Records and Documentation for Excess Reagents
Examples of Regulatory Agencies Enforcing Standards for Managing Excess Reagents
Best Practices for Implementing a Laboratory Inventory System
Designing an Effective Inventory System
Establishing a System for Tracking and Monitoring Chemical Inventory
Maintaining an Accurate and Up-to-Date Inventory
Managing Excess Reagents in a Sustainable and Environmentally Friendly Manner
Designing a Sustainable Waste Management Program
Examples of Laboratories with Sustainable Practices
Benefits of Sustainable Waste Management, How to find reagent in excess
Benefits of Sustainable Waste Management
Examples
Reduced environmental impact
LBNL’s Zero-Waste policy has reduced waste generation by 90% since its implementation.
Cost savings
The University of California, Berkeley’s College of Chemistry has saved $100,000 annually by implementing a waste reduction program.
Improved regulatory compliance
LBNL’s Zero-Waste policy has improved the laboratory’s compliance with environmental regulations, reducing the risk of fines and penalties.
Ultimate Conclusion

Top FAQs: How To Find Reagent In Excess