As how to find the pH value takes center stage, this opening passage beckons readers into a world crafted with good knowledge, ensuring a reading experience that is both absorbing and distinctly original.
Understanding the pH value is crucial in various environments, including oceans, rivers, and lakes. The pH level affects the survival of aquatic plants and animals, with specific species requiring certain pH levels to thrive.
Understanding the Importance of pH in Various Environments
pH plays a crucial role in maintaining ecological balance in different ecosystems, including oceans, rivers, and lakes. The pH level of an ecosystem can affect the survival and diversity of aquatic plants and animals, making it essential to understand the importance of pH in various environments. Water pH affects the availability of essential nutrients, such as calcium and magnesium, which are crucial for the growth and development of aquatic life.
The pH level of an ecosystem can range from highly acidic (pH 0-3) to alkaline (pH 9-14). Different aquatic species have adapted to thrive in these various pH ranges. For instance, acidophilic organisms, such as certain species of bacteria and archaea, can survive and even thrive in acidic environments with pH levels as low as 0.5. On the other hand, alkaliphilic organisms, such as some species of cyanobacteria and green algae, can tolerate and even require highly alkaline conditions with pH levels above 9.
Differential pH Requirements for Aquatic Life
The pH requirements for aquatic life vary significantly depending on the species. Some aquatic species are highly sensitive to pH changes and can be affected even by slight changes in pH levels. For example, some species of fish, such as the salmon, require a relatively narrow pH range to survive. Changes in pH levels can also affect the availability of essential nutrients, such as calcium and magnesium, which are crucial for the growth and development of aquatic life.
Examples of Aquatic Species Affected by pH Changes
Aquatic plants and animals have varying levels of tolerance to pH changes. Some species can survive and thrive in a wide range of pH levels, while others are highly sensitive to pH changes. For example:
- Acidophilic organisms, such as certain species of bacteria and archaea, can survive and even thrive in acidic environments with pH levels as low as 0.5.
- Alkaliphilic organisms, such as some species of cyanobacteria and green algae, can tolerate and even require highly alkaline conditions with pH levels above 9.
- Some species of fish, such as the salmon, require a relatively narrow pH range to survive and are highly sensitive to changes in pH levels.
- Certain species of shellfish, such as oysters and mussels, require a specific pH range to survive and are highly sensitive to changes in pH levels.
Impacts of pH Changes on Aquatic Ecosystems
Changes in pH levels can have significant impacts on aquatic ecosystems. For example, a sudden increase in pH levels can lead to the release of excess carbon dioxide from the water, which can cause a buildup of toxic gases in the water column. Similarly, a sudden decrease in pH levels can lead to the release of excess hydrogen ions, which can cause a buildup of acid in the water column. These changes can lead to a decline in aquatic biodiversity and even extinctions.
Aquatic ecosystems have a delicate balance of pH levels, and changes in pH levels can have far-reaching consequences for aquatic life. Understanding the importance of pH in various environments is crucial for maintaining ecological balance and ensuring the survival and diversity of aquatic species. The pH level of an ecosystem can range from highly acidic (pH 0-3) to alkaline (pH 9-14), and different aquatic species have adapted to thrive in these various pH ranges.
Calculating pH
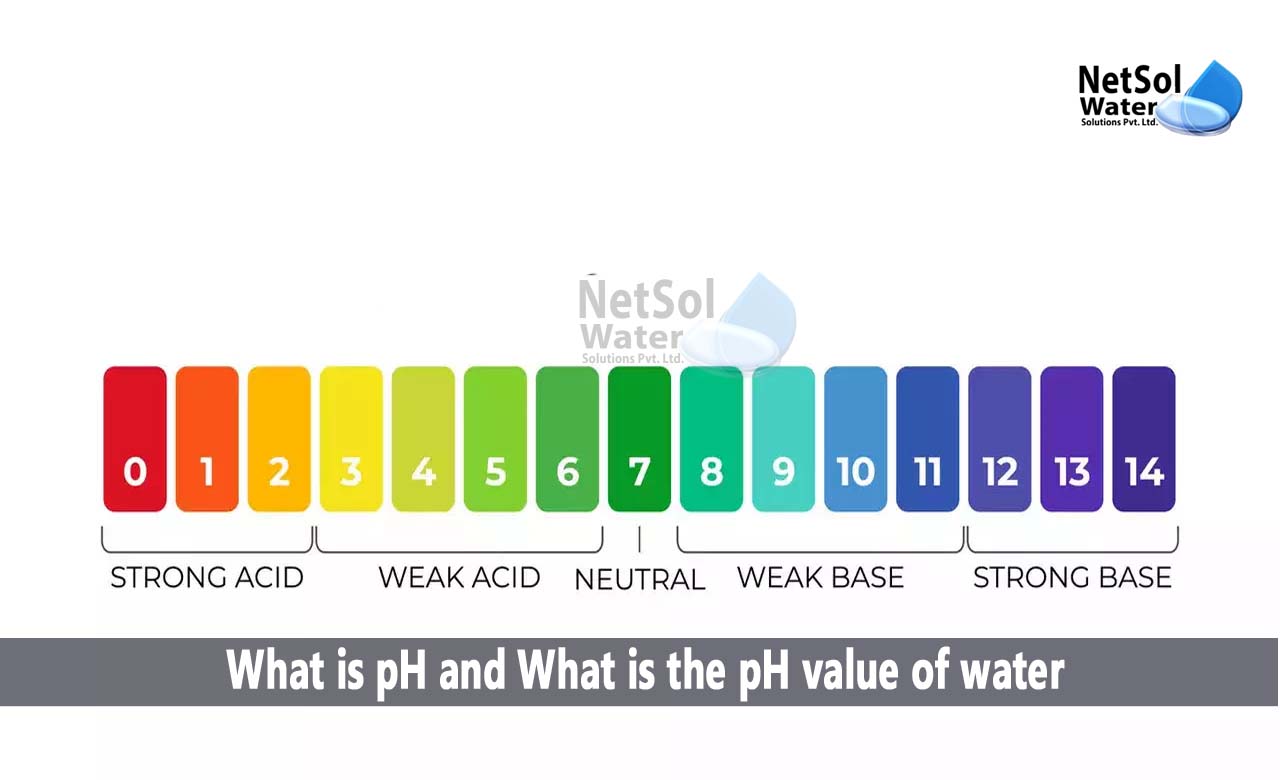
Calculating the pH of a solution is essential in understanding its acidity or alkalinity. Various methods can be employed to determine the pH, including the use of pH tables, pH meters, and the Henderson-Hasselbalch equation. In this section, we will delve into the Henderson-Hasselbalch equation and its application in calculating pH.
The Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation is a mathematical formula used to calculate the pH of a buffer solution. A buffer solution is a mixture of a weak acid and its conjugate base, or a weak base and its conjugate acid. The equation is given by:
pH = pKa + log_10 ([A^-]/[HA])
Where pKa is the acid dissociation constant of the weak acid, [A^-] is the concentration of the conjugate base, and [HA] is the concentration of the weak acid.
pKa = -log_10 (Ka)
Where Ka is the acid dissociation constant.
Step-by-Step Guide to Using the Henderson-Hasselbalch Equation
To use the Henderson-Hasselbalch equation, follow these steps:
- Identify the weak acid and its conjugate base in the buffer solution.
- Determine the pKa of the weak acid from the acid dissociation constant (Ka).
- Measure the concentrations of the conjugate base and the weak acid in the solution.
- Plug the values into the Henderson-Hasselbalch equation to calculate the pH of the buffer solution.
Types of Buffers and Their pH-Stability Ranges, How to find the ph value
There are several types of buffers, each with its own pH-stability range. Some common types of buffers include:
- Acidic Buffers: These buffers have a pH-stability range of 3.0-5.0 and are commonly used in biological systems. Examples include phosphoric acid and acetic acid.
- Neutral Buffers: These buffers have a pH-stability range of 5.0-8.0 and are commonly used in biochemistry and molecular biology. Examples include phosphate buffer and Tris buffer.
- Basic Buffers: These buffers have a pH-stability range of 8.0-10.0 and are commonly used in biochemistry and molecular biology. Examples include tris(hydroxymethyl)aminomethane (Tris) buffer and glycine buffer.
Comparison of pH Calculation Methods
There are several methods to calculate pH, each with its own strengths and weaknesses. Some common methods include:
- pH Tables: pH tables are pre-calculated tables that provide the pH of solutions containing a certain concentration of acids or bases. This method is quick and easy but limited in its accuracy.
- pH Meters: pH meters are electronic devices that measure the pH of a solution directly. This method is highly accurate and reliable but can be expensive and require calibration.
- Henderson-Hasselbalch Equation: The Henderson-Hasselbalch equation is a mathematical formula used to calculate the pH of a buffer solution. This method is accurate and reliable but requires knowledge of the acid dissociation constant and concentrations of the acid and base.
pH Indicators
pH indicators are substances that change color in response to changes in the pH of a solution, allowing us to determine the acidity or alkalinity of a substance. These indicators are crucial in various fields, including chemistry, biology, and medicine, where precise pH measurements are essential for understanding chemical reactions, biological processes, and medical treatments.
Properties and Characteristics of pH Indicators
pH indicators exhibit unique properties that make them useful for pH measurement. The color-changing ranges of pH indicators depend on the specific compound and the pH environment. Generally, pH indicators change color at a specific pH range, which depends on their molecular structure. For example, litmus changes color at pH 4.5 and 8.3, while phenolphthalein changes color at pH 8.3 or higher. Some pH indicators also exhibit stability in various solvents, including water, alcohol, and acetic acid.
Common pH Indicators and their Applications
Below are some common pH indicators and their applications in various fields:
- Litmus: A common natural pH indicator used in chemistry and biology experiments to determine pH levels.
- Phenolphthalein: A synthetic pH indicator used in chemistry and medicine to measure pH levels in weakly acidic or basic solutions.
- Methyl orange: A synthetic pH indicator used in chemistry and biology experiments to determine pH levels in weakly acidic solutions.
- Alizarin: A natural pH indicator used in chemistry and biology experiments to determine pH levels in strongly basic solutions.
- Thymol blue: A synthetic pH indicator used in medicine to measure pH levels in blood and other biological fluids.
- Cresol red: A synthetic pH indicator used in chemistry and biology experiments to determine pH levels in strong acidic and basic solutions.
Comparison of pH Indicators
When selecting a pH indicator for a particular application, it is essential to consider the advantages and disadvantages of each option. The following table summarizes the characteristics of common pH indicators:
| Indicator | Color- Changing Range | Stability | Cost | Availability |
|---|---|---|---|---|
| Litmus | pH 4.5 and 8.3 | Unstable in strong acids and bases | Low | Readily available |
| Phenolphthalein | pH 8.3 or higher | Stable in weakly acidic and basic solutions | Medium | Commonly available |
| Methyl orange | pH 3.1 and 4.4 | Unstable in strong acids and bases | Low | Readily available |
When selecting a pH indicator, it is crucial to consider the pH range, stability, cost, and availability of the indicator.
pH Buffering Systems
pH buffering systems are essential in maintaining acid-base homeostasis in living organisms. These systems allow the regulation of pH levels within narrow ranges, preventing excessive acidity or alkalinity that can damage tissues and disrupt cellular function. The buffering capacity of a solution is its ability to resist changes in pH when acids or bases are added.
Understanding acid-base equilibria is crucial in the context of pH buffering systems. This concept describes the dynamic equilibrium between an acid and its conjugate base, or between a base and its conjugate acid. For example,
HA ⇌ H+ + A−
represents the equilibrium between a weak acid (HA) and its conjugate base (A−). The equilibrium constant (Ka) for this reaction determines the acid’s strength and its ability to donate H+ ions.
### Types of pH Buffering Systems
#### Biological pH Buffers
Biological pH buffers play a critical role in maintaining acid-base homeostasis in living organisms. These buffers include proteins such as hemoglobin and albumin, which can bind and buffer H+ ions. Other examples of biological pH buffers include bicarbonate (HCO3−) / carbonic acid (H2CO3) buffer in the blood and phosphate buffer in cells.
#### Chemical pH Buffers
Chemical pH buffers are man-made solutions designed to resist changes in pH. Common examples of chemical pH buffers include phosphate buffers, Tris buffers, and borate buffers. These buffers can be used in various applications, such as in cell cultures and biochemistry experiments.
### Mechanisms of pH Buffering
pH buffering systems work by reducing the amount of free H+++3−) reacts with the H+2CO3), which then dissociates to water and carbon dioxide. This reaction effectively reduces the concentration of free H++−+3−) and the subsequent formation of carbonic acid (H2CO3) allow the blood to maintain a stable pH level between 7.35 and 7.45. This process is critical for the proper functioning of the brain, nervous system, and other organs that are sensitive to changes in pH.
Similarly, pH buffering systems in cells, such as the phosphate buffer system, play a crucial role in maintaining intracellular pH homeostasis. This is essential for proper cellular function, as changes in pH can affect enzyme activity, protein structure, and other cellular processes.
Final Review: How To Find The Ph Value
In conclusion, finding the pH value requires careful consideration of the environment, measurement tools, and calculation methods. By understanding the pH value, we can maintain ecological balance and ensure the survival of aquatic life.
FAQs
What is the ideal pH range for human blood?
The ideal pH range for human blood is between 7.35 and 7.45.
How often should I calibrate my pH meter?
It is recommended to calibrate your pH meter before each use, as well as at regular intervals to ensure accuracy.
Can pH indicators be used to measure pH levels in food?
Yes, pH indicators can be used to measure pH levels in food, but they may not be as accurate as other methods. Always follow proper food safety guidelines when using pH indicators in food.
What is the difference between acidic and alkaline foods?
Acidic foods, such as citrus fruits, have a lower pH level than alkaline foods, such as spinach, which have a higher pH level.