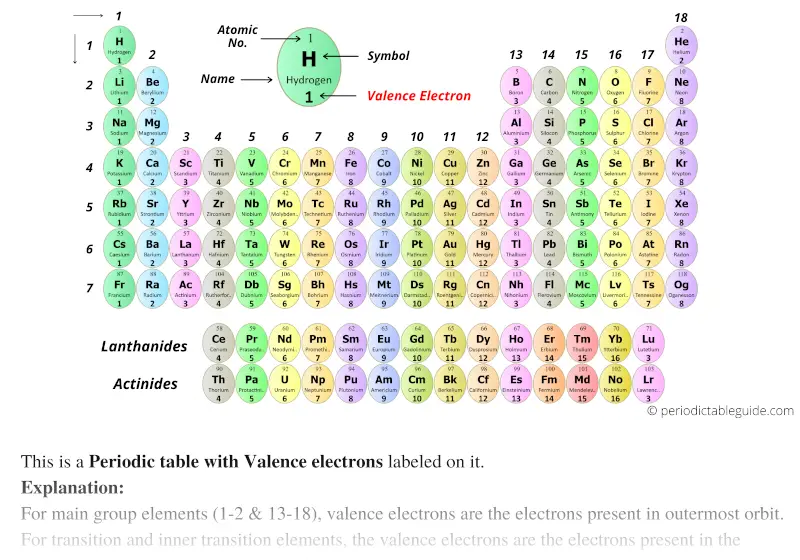
Kicking off with how to find valence electrons, this journey unlocks the secrets of chemical bonding and reactivity. Valence electrons are the building blocks of atoms, and understanding them is crucial in predicting the behavior of elements in different compounds. With the right approach, finding valence electrons becomes an essential skill for chemistry enthusiasts and professionals alike.
From the periodic table to Lewis structures, and from VSEPR theory to exceptions to the octet rule, this comprehensive guide will walk you through the methods and trends that will help you find valence electrons. You’ll learn how to determine valence electrons, how to visualize them in molecules, and how to apply this knowledge to real-world applications.
Valence Electrons: An Essential Component of Chemistry
Valence electrons play a crucial role in understanding the properties and behavior of elements in chemistry. They are responsible for determining the chemical reactivity and bonding capabilities of an element, which is essential in understanding various chemical processes and reactions.
Valence electrons are the electrons present in the outermost shell of an atom, and they are responsible for forming chemical bonds with other atoms. The number of valence electrons in an atom determines its valency, which is the number of bonds it can form with other atoms. Elements with low valence electron counts tend to be more reactive, while those with high valence electron counts tend to be less reactive.
Examples of Elements and Their Valence Electrons
The valence electrons of various elements can be understood through examples. For instance, hydrogen has one valence electron, which makes it highly reactive. Carbon has four valence electrons, which allows it to form stable bonds with other elements. Oxygen has six valence electrons, which makes it highly reactive and capable of forming multiple bonds with other elements. On the other hand, Neon has a full outer shell with eight valence electrons, which makes it stable and unreactive.
Here are some examples of elements and their valence electrons:
- Hydrogen (H): 1
- Carbon (C): 4
- Oxygen (O): 6
- Neon (Ne): 8
Valence Electrons and Chemical Bonding
The valence electrons of an element determine its chemical bonding capabilities. For example, when hydrogen forms a bond with another hydrogen atom, it shares its valence electron to form a covalent bond. When oxygen forms a bond with hydrogen, it shares its two valence electrons to form a covalent bond.
Valence electrons are the key to understanding chemical bonding and reactivity.
Valence Electrons Trends in the Periodic Table
The first 20 elements in the periodic table have unique trends and patterns when it comes to valence electrons. These trends can be summarized as follows:
- As we move from left to right in a period, the number of valence electrons increases.
- As we move down a group, the number of valence electrons increases.
- Elements in the s-block have one or two valence electrons.
- Elements in the p-block have three to six valence electrons.
Valence Electrons Table for the First 20 Elements
| Element | Atomic Number | Valence Electrons |
|---|---|---|
| Hydrogen (H) | 1 | 1 |
| Helium (He) | 2 | 2 |
| Lithium (Li) | 3 | 1 |
| Beryllium (Be) | 4 | 2 |
| Boron (B) | 5 | 3 |
| Carbon (C) | 6 | 4 |
| Nitrogen (N) | 7 | 5 |
| oxygen (O) | 8 | 6 |
| Fluorine (F) | 9 | 7 |
| Neon (Ne) | 10 | 8 |
| Sodium (Na) | 11 | 1 |
| Magnesium (Mg) | 12 | 2 |
| Aluminum (Al) | 13 | 3 |
| Silicon (Si) | 14 | 4 |
| Phosphorus (P) | 15 | 5 |
| Sulfur (S) | 16 | 6 |
| Chlorine (Cl) | 17 | 7 |
| Argon (Ar) | 18 | 8 |
| Potassium (K) | 19 | 1 |
| Calcium (Ca) | 20 | 2 |
Visualizing Valence Electrons
Visualizing valence electrons is a crucial step in understanding the properties and behavior of molecules. By representing the valence electrons in a diagram, we can gain insight into the molecular structure and how atoms interact with each other. This can be particularly useful when studying chemical reactions, bonding, and molecular stability.
In this section, we will explore the valence electrons of different molecules through illustrated diagrams and detailed explanations.
The Valence Electrons in Water (H2O)
The water molecule consists of two hydrogen atoms and one oxygen atom. Oxygen has six valence electrons, and each hydrogen atom has one valence electron. The valence electrons in water can be represented as follows:
Oxygen (O): 2s² 2p⁴
The oxygen atom shares its two valence electrons with the two hydrogen atoms, forming covalent bonds. Each hydrogen atom shares its one valence electron with the oxygen atom. The valence electrons in water can be visualized as the following:
Atom Valence Electrons Oxygen (O) 6 (2 electrons with H1 + 2 electrons with H2) Hydrogen (H1) 2 (1 electron shared with O) Hydrogen (H2) 2 (1 electron shared with O)
The Valence Electrons in Ammonia (NH3), How to find valence electrons
The ammonia molecule consists of one nitrogen atom and three hydrogen atoms. Nitrogen has five valence electrons, and each hydrogen atom has one valence electron. The valence electrons in ammonia can be represented as follows:
Nitrogen (N): 2s² 2p³
The nitrogen atom shares three of its valence electrons with the three hydrogen atoms, forming covalent bonds. Each hydrogen atom shares its one valence electron with the nitrogen atom. The valence electrons in ammonia can be visualized as the following:
Atom Valence Electrons Nitrogen (N) 8 (3 electrons with H1 + 2 electrons with H2 + 3 electrons with H3) Hydrogen (H1) 2 (1 electron shared with N) Hydrogen (H2) 2 (1 electron shared with N) Hydrogen (H3) 2 (1 electron shared with N)
The Valence Electrons in Methane (CH4)
The methane molecule consists of one carbon atom and four hydrogen atoms. Carbon has four valence electrons, and each hydrogen atom has one valence electron. The valence electrons in methane can be represented as follows:
Carbon (C): 2s² 2p²
The carbon atom shares four of its valence electrons with the four hydrogen atoms, forming covalent bonds. Each hydrogen atom shares its one valence electron with the carbon atom. The valence electrons in methane can be visualized as the following:
Atom Valence Electrons Carbon (C) 8 (4 electrons with H1 + 2 electrons with H2 + 2 electrons with H3) Hydrogen (H1) 2 (1 electron shared with C) Hydrogen (H2) 2 (1 electron shared with C) Hydrogen (H3) 2 (1 electron shared with C) Hydrogen (H4) 2 (1 electron shared with C)
Understanding Exceptions to the Octet Rule
The octet rule is a fundamental principle in chemistry that states atoms tend to gain, lose, or share electrons to achieve a full outer energy level, resulting in a stable electronic configuration with eight valence electrons. However, this rule is not universally applicable, and there are exceptions that deviate from this stability.
Many elements exhibit a stable electronic configuration with fewer than eight valence electrons, often due to the absence of electrons in the outermost energy level. For example, helium (He) has a closed outer energy level with two valence electrons, which is stable. Similarly, hydrogen (H) has only one valence electron, which is sufficient for its atomic stability.
Examples of Stable Compounds that Deviate from the Octet Rule
Stable compounds that deviate from the octet rule often have unique structures or compositions that enable them to achieve stability with fewer or more than eight valence electrons. These compounds play crucial roles in various chemical reactions and are essential components of many natural processes.
Some notable examples of stable compounds that deviate from the octet rule include:
- Hydrogen Fluoride (HF)
- Phosphorus Trichloride (PCl3)
- Phosphorus Pentachloride (PCl5)
These compounds exhibit stability with fewer than eight valence electrons on the central atom. For instance, in hydrogen fluoride (HF), the fluorine atom shares its lone pair of electrons with the hydrogen atom, resulting in a stable molecule with six valence electrons on the fluorine atom.
Bonding in HF: H ∙∙∙ F:σ (1s)²²
Additionally, some compounds exhibit stability with more than eight valence electrons on the central atom. For example, xenon tetrafluoroplatinate (XePtF6), also known as xenon hexafluoroplatinate (Xe[PtF6]−), has a central xenon atom bonded to six fluorine atoms, resulting in a stable compound with ten valence electrons on the xenon atom.
Bonding in XePtF6: Xe ∙∙∙ Pt (F)₆:σ (4d)¹⁴²
These examples illustrate the diversity of stable chemical compounds that deviate from the octet rule, highlighting the complexity and nuance of chemical bonding.
Importance of Exceptions to the Octet Rule
The octet rule, while a fundamental principle in chemistry, has its limitations. Exceptions to the octet rule highlight the versatility and adaptability of chemical bonding, allowing for the formation of a wide range of stable compounds with unique properties and structures.
Understanding these exceptions is crucial for predicting and manipulating chemical reactions, designing new materials and compounds, and explaining the behavior of atoms and molecules in various environments. By embracing the complexities of the octet rule and its exceptions, we can deepen our comprehension of the molecular world and unlock new breakthroughs in chemistry and related fields.
Visualizing Valence Electrons in Real-World Applications
The structure of valence electrons in molecules is a crucial aspect of understanding chemical properties and reactivity. In this section, we’ll explore the real-world implications of visualizing valence electrons and their importance in various fields.
In the study of chemistry, it’s often said that the arrangement of valence electrons in molecules mirrors the real-world structure of proteins, DNA, and other biomolecules. This analogy is not just a metaphor; it provides valuable insight into the underlying chemical bonds that govern the behavior of these complex molecules.
Arrangement of Valence Electrons in Proteins
Proteins are biological molecules composed of amino acids, each with a unique arrangement of valence electrons. The sequence of amino acids determines the three-dimensional structure of a protein, which in turn influences its function and interactions with other molecules. For instance, enzymes, a class of proteins, rely on the precise arrangement of valence electrons to catalyze chemical reactions.
- Enzymes use their valence electrons to form covalent bonds with reactants, facilitating the conversion of substrates into products.
- The spatial arrangement of valence electrons in enzymes enables selective catalysis, allowing them to target specific reactions and molecules.
- Understanding the valence electron arrangement in proteins has led to the development of targeted therapies, where enzymes are engineered to bind specifically to disease-causing molecules.
Valence Electrons in DNA
DNA (Deoxyribonucleic acid) is a molecule that contains the genetic instructions used in the development and function of all living organisms. The double helix structure of DNA is stabilized by hydrogen bonds, which are formed between valence electrons of the nitrogenous bases that make up the molecule.
- The arrangement of valence electrons in the nitrogenous bases (adenine, guanine, cytosine, and thymine) allows DNA to store genetic information in a stable and accessible way.
- Understanding the valence electron arrangement in DNA has enabled the development of gene editing technologies like CRISPR, which rely on precise interactions with specific sequences of DNA.
- Studying the valence electrons in DNA has shed light on the mechanisms of genetic diseases, such as mutations and epigenetic changes that can lead to cancer.
Importance of Understanding Valence Electrons in Applications
The study of valence electrons is crucial in various fields, including organic synthesis and pharmaceutical development. Understanding the arrangement of valence electrons in molecules allows chemists to predict and design the properties of new compounds, which can lead to breakthroughs in medicine and materials science.
- Accurate predictions of valence electron arrangement enable the design of new medications with improved potency and selectivity, reducing side effects and improving treatment outcomes.
- Understanding valence electron arrangement in molecules is essential for the development of new materials with unique properties, such as conductivity, strength, or optical behavior.
- Knowledge of valence electron arrangement has led to the creation of new catalysts and reagents in organic synthesis, facilitating the development of complex molecules and pharmaceuticals.
Closing Notes
With a solid understanding of how to find valence electrons, you’ll be well-equipped to tackle the complexities of chemical bonding and reactivity. This knowledge will open doors to new discoveries and insights, helping you to better understand the intricacies of molecular structures and interactions. So, embark on this journey and unlock the secrets of valence electrons today!
Question Bank: How To Find Valence Electrons
What are valence electrons, and why are they important?
Valence electrons are the electrons in the outermost energy level of an atom, and they play a crucial role in chemical bonding and reactivity. Understanding valence electrons is essential for predicting the behavior of elements in different compounds and for designing new materials and molecules.
How do I determine the valence electrons of an atom?
You can determine the valence electrons of an atom by using the periodic table, Lewis structures, and VSEPR theory. The number of valence electrons an atom has depends on its position in the periodic table and its electron configuration.
What is the octet rule, and why is it important?
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer energy level with eight electrons. This rule is important because it helps us understand how atoms form chemical bonds and how to design molecules with specific properties.
Can valence electrons be visualized in molecules?
Yes, valence electrons can be visualized in molecules using Lewis structures and other molecular representations. Visualizing valence electrons helps us understand the molecular structure and behavior of a compound.