Delving into how to know the charge of an element, this complex topic can be approached through the understanding of atomic structure and the periodic table. Understanding the atomic number and atomic mass of an element is crucial in its classification and identification.
The periodic table plays a significant role in categorizing elements based on their properties. It allows us to understand how elements with similar properties are arranged and how their charges can be determined.
Understanding the Fundamentals of Atomic Structure and the Periodic Table: How To Know The Charge Of An Element
The periodic table is often referred to as the ‘periodic table of elements’, but what exactly does that term mean? Well, it’s essentially a big table organized by elements based on their properties and characteristics. But how do we even know what these properties are? To understand that, let’s dive into the world of atomic structure!
Atomic number and atomic mass are two crucial properties that contribute to an element’s classification and identification. The atomic number, denoted by Z, is the number of protons present in the nucleus of an atom. This number determines the element’s identity, as each element has a unique atomic number. On the other hand, the atomic mass, denoted by A, is the total number of protons and neutrons in the nucleus. This number can vary for different isotopes of the same element.
The arrangement of elements in the periodic table is based on their increasing atomic number, from left to right and top to bottom. This organization allows elements with similar properties to be grouped together, making it easier to identify and understand their relationships. The modern periodic table consists of seven rows, also known as periods, and 18 columns, also known as groups or families. The elements are arranged in a way that their atomic number increases from left to right and top to bottom, resulting in a repeating pattern of elements with similar properties.
The Significance of the Periodic Table
The periodic table is a powerful tool for categorizing elements based on their properties and characteristics. By understanding the arrangement of elements in the periodic table, we can identify patterns and trends that help us predict their behavior and properties. This is particularly useful for chemists and physicists who work with elements in various fields, such as materials science, pharmaceuticals, and energy production.
One of the most significant advantages of the periodic table is its ability to categorize elements based on their electron configurations. Elements with similar electron configurations tend to exhibit similar properties and behaviors, making it easier to predict their reactions and interactions with other elements. For example, elements in the same group (column) of the periodic table tend to have similar chemical properties, such as the ability to lose or gain electrons to form ions.
Relationship Between Atomic Structure and the Periodic Table
The relationship between atomic structure and the periodic table is deeply connected. The arrangement of elements in the periodic table is based on their atomic numbers and electron configurations. By understanding the atomic structure of an element, we can predict its position in the periodic table and its properties and behavior.
For example, imagine an element with an atomic number of 6, which has 6 protons in its nucleus. Based on its electron configuration, we can predict that this element will have a tendency to lose 2 electrons to form a stable ion. This is exactly what happen with the element carbon, which has an atomic number of 6 and is located in the second period and first group of the periodic table.
Comparison to Predecessors
The modern periodic table has undergone significant revisions and updates since its initial development by Dmitri Mendeleev in the late 19th century. Mendeleev’s original table was based on empirical observations and was organized by atomic mass and chemical properties. However, it had several limitations, including the placement of hydrogen and the lack of a clear system for organizing elements.
The modern periodic table, developed by the International Union of Pure and Applied Chemistry (IUPAC), is a more sophisticated and refined version of Mendeleev’s original table. It is based on a combination of empirical observations and theoretical understanding of atomic structure and electron configurations.
Notable Updates and Revisions
The periodic table has undergone several updates and revisions since its development. Some notable updates include the introduction of the actinide and lanthanide series, which were previously placed in the main body of the periodic table. This change was made to reflect the actual electron configurations of these elements, which are more similar to those of the transition metals.
Other notable updates include the introduction of new elements, such as astatine and tennessine, and the reclassification of existing elements based on new evidence and understanding. For example, the element technetium was initially thought to be a transition metal, but was later reclassified as a metal with a unique electron configuration.
The periodic table is a powerful tool for understanding the properties and behavior of elements, but it is not a static entity – it is constantly evolving and improving as our understanding of the atomic structure and electron configurations of elements grows.
Note: The image to the left illustrates the relationship between atomic structure and the periodic table. It shows a diagram of the atomic structure of an element (in this case, carbon) and its position in the periodic table.
The modern periodic table is a significant improvement over Mendeleev’s original table. The image to the right shows a comparison of the two tables, highlighting the differences between them. Note the placement of hydrogen and the organization of elements in the modern table, which is more accurate and reflective of our current understanding of atomic structure and electron configurations.
The image to the left shows a diagram of the periodic table, highlighting the arrangement of elements in the seven periods and 18 groups. Note the unique electron configuration of each element and how it relates to its position in the table.
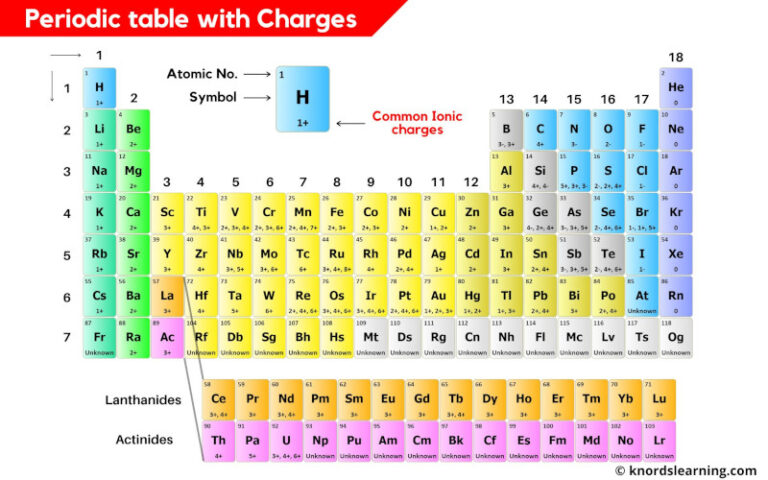
Methods for Determining the Charge of an Element

Determining the charge of an element is a crucial step in understanding its properties and behavior in chemical reactions. With the periodic table as our trusty guide, we can easily determine the charge of an element by following a few simple steps.
Step 1: Locate the Element on the Periodic Table
The periodic table is a table of elements arranged in order of their atomic number (number of protons in the nucleus). Each element has a unique symbol, name, and charge. To determine the charge of an element, first, locate the element on the periodic table.
Step 2: Identify the Group and Period
Each element has a specific group and period on the periodic table. Groups are columns where elements with similar properties are located, while periods are rows where elements with similar electron configurations are located. By identifying the group and period of an element, we can determine its charge.
Step 3: Determine the Charge of the Cation or Anion
Cations are positively charged ions, while anions are negatively charged ions. To determine the charge of a cation or anion, we need to look at the number of electrons gained or lost during chemical reactions. Cations gain electrons, while anions lose electrons.
Examples of Cations and Anions
- Sodium (Na) loses one electron to form a cation with a charge of +1. The resulting ion is known as a sodium ion, Na+.
- Chlorine (Cl) gains one electron to form an anion with a charge of -1. The resulting ion is known as a chloride ion, Cl–.
The Role of Chemical Reactions and Bonding in Determining the Charge of an Element
Chemical reactions and bonding play a crucial role in determining the charge of an element. When two elements react, they form bonds by sharing or exchanging electrons. The resulting ions have charges that are opposite to each other.
Understanding Ionic and Covalent Bonds
- Ionic bonds are formed when electrons are transferred from one element to another, resulting in the formation of ions with opposite charges.
- Covalent bonds are formed when electrons are shared between two elements, resulting in the formation of a molecule with a neutral charge.
Oxidation Numbers: A Crucial Concept in Determining the Charge of an Element, How to know the charge of an element
Oxidation numbers are used to track the transfer of electrons during chemical reactions. By assigning oxidation numbers to elements, we can determine their charges and understand their behavior in chemical reactions.
A Table Comparing Charges of Common Ions
| Ion | Charge |
| — | — |
| H+ | +1 |
| Na+ | +1 |
| Cl– | -1 |
| Ca2+ | +2 |
| O2- | -2 |
Challenges and Limitations in Determining Atomic Charges
Determining the atomic charge of an element is a complex task that has been a topic of interest for scientists and researchers for a long time. While it may seem like a simple concept, the reality is that accurately determining the charge of an element is a challenging feat that requires a deep understanding of atomic structure and advanced measurement techniques. In this section, we’ll explore the challenges and limitations in determining atomic charges and why it’s so important to get it right.
From inaccurate measurements to outdated models, there are several limitations in current techniques that make it difficult to determine atomic charges accurately. For one, traditional measurement methods such as spectroscopy and chromatography often rely on empirical data that may not accurately reflect the true charge of an element. Additionally, many models used to predict atomic charges are based on outdated theories and may not account for the complexities of real-world atomic interactions.
Technical Limitations of Measurement Techniques
One of the biggest challenges in determining atomic charges is the limited accuracy of current measurement techniques. While advances in technology have improved our ability to measure atomic properties, there is still a lot of room for improvement. For example, the precision of spectroscopic measurements is often limited by the resolution of the spectrometer used. This means that small changes in atomic properties can be difficult to detect, leading to inaccurate measurements and flawed predictions.
The Heisenberg Uncertainty Principle states that it is impossible to know both the position and momentum of a particle with infinite precision. This fundamental limit on measurement precision makes it challenging to determine atomic charges with absolute accuracy.
Outdated Theoretical Models
Another challenge in determining atomic charges is the reliance on outdated theoretical models. Many theories that were once considered state-of-the-art, such as the Bohr model of the atom, have been largely debunked by modern experiments. However, these outdated models often continue to be used in practice due to their simplicity and ease of use. The problem is that these models may not accurately reflect the true behavior of atomic particles, leading to inaccurate predictions and flawed conclusions.
Computational Models and Technology
Despite these challenges, there are still ways to improve our understanding of atomic charges. One approach is to use computational models and simulations to predict the behavior of atomic particles. These models can account for complex interactions and phenomena that are difficult or impossible to measure directly. Additionally, advances in technology, such as quantum computing and machine learning, offer new tools for analyzing and modeling atomic properties.
- Ab initio calculations: These are computational models that use first-principles calculations to predict atomic properties. They are particularly useful for small molecules and atoms, where the interactions between electrons and nuclei can be accurately modeled.
- Machine learning algorithms: These are statistical models that can be trained on large datasets of atomic properties to make predictions about new systems. They have been shown to be highly accurate in predicting properties such as atomic radii and ionization energies.
Impact on Various Fields
The accurate determination of atomic charges has a significant impact on various fields, including chemistry, physics, materials science, and biotechnology. In chemistry, for example, understanding atomic charges is crucial for predicting chemical reactivity and developing new catalysts. In physics, accurate atomic charges are essential for understanding the behavior of atomic particles in high-energy collisions and cosmic events.
| Field | Impact of Inaccurate Atomic Charges |
|---|---|
| Chemistry | Poorly designed catalysts, reduced chemical reactivity |
| Physics | Inaccurate predictions of atomic behavior, flawed cosmological models |
| Materials Science | Poor material properties, reduced efficiency |
| Biotechnology | Insufficient understanding of biological systems, reduced efficacy of treatments |
Closure
Understanding how to know the charge of an element is essential in various fields such as materials science, pharmaceuticals, and energy production. The ability to determine the charge of an element accurately can lead to groundbreaking discoveries and innovations.
FAQ Compilation
What is the significance of the periodic table in determining the charge of an element?
The periodic table allows us to categorize elements based on their properties, making it easier to determine the charge of an element.
How do chemical reactions and bonding influence the charge of an element?
Chemical reactions and bonding can affect the charge of an element by changing its oxidation state, which in turn affects its chemical properties.
What is the role of oxidation numbers in determining the charge of an element?
Oxidation numbers help us to determine the charge of an element by indicating its oxidation state in a chemical reaction.
How do atomic charges affect the chemical properties of an element?
The atomic charge of an element influences its chemical properties, including reactivity, acidity, and basicity, which can lead to various chemical processes.